December 31, 2020 — After “COVID-19,” the term that most people will remember best from 2020 is likely to be “social ...
Coronavirus (COVID-19)
This page contains medical information for clinicians on the 2019 Novel Coronavirus (COVID-19, also called 2019-nCoV and now clinically SARS‐CoV‐2). This section includes articles on medical imaging of the virus for radiologists, new technologies being deployed to fight the virus and clinical information from various sources. Here are direct links for medical professionals to COVID-19 resources from the U.S. Food and Drug Administration (FDA), Centers for Disease Control (CDC) and the World Health Organization (WHO). Daily world-wide statistics on the coronavirus outbreak are available from the WHO Situations Reports. Centers for Medicare and Medicaid Services (CMS) frequently asked questions and answers (FAQs) for healthcare providers regarding Medicare payment for laboratory tests and other services related to the COVID-19
Here are the top 25 best performing articles on the Imaging Technology News (ITN) website from 2020. This is based on ...
Here are the top 25 best performing videos on the Imaging Technology News website (ITN) from the past year, based on ...
Members of the enterprise imaging technology community are facing significant changes in the market, due to the COVID-19 ...
December 23, 2020 — The U.S. Food and Drug Administration December 18 issued an emergency use authorization (EUA) for a ...
December 21, 2020 — According to recent studies, an increase in the COVID-19 positivity rate correlates to an increase ...
December 21, 2020 — The Radiological Society of North America (RSNA) and the RSNA COVID-19 AI Task Force announced that ...
The COVID-19 pandemic accelerated a change that was already occurring in imaging-heavy practices across the country — ...
December 14, 2020 — The U.S. Food and Drug Administration (FDA) issued the first emergency use authorization (EUA) for ...
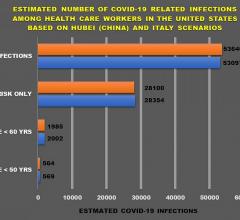
December 16, 2020 — During the COVID-19 pandemic, health care workers have been at the forefront of the battle against ...
December 11, 2020 — For some, COVID-19 can result in severe pneumonia or even death, while others remain purely ...
The coronavirus pandemic has changed the way radiologists read images and today facilities struggle to define a “new ...
December 8, 2020 — After recovering from COVID-19, some patients are left with chronic, debilitating pain, numbness or ...
December 7, 2020 — An analysis of lung tissues from patients with different types of pulmonary fibrosis - including ...
December 7, 2020 — The U.S. Food and Drug Administration (FDA) is informing patients and health care providers that ...
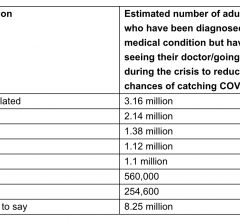
December 7, 2020 — New research* from Zegami, an Oxford based medical image analysis platform, reveals that 29% of ...
December 4, 2020 — Today, the U.S. Food and Drug Administration (FDA) issued an immediately in effect guidance ...
December 3, 2020 — Northwestern University researchers have developed a new artificial intelligence (AI) platform that ...
December 1, 2020 — As hospitals see an influx of COVID-19 (SARS-CoV-2) patients, many are looking for ways to rapidly ...
December 1, 2020 — Visage Imaging, Inc. ("Visage"), a wholly owned subsidiary of Pro Medicus Ltd. (ASX: PME), has ...
November 30, 2020 — AI Medic Inc. announced that it has obtained official product certification from the NIDS (National ...
November 30, 2020 — GE Healthcare unveiled a slate of new intelligently efficient solutions to help clinicians solve ...
November 29, 2020 — Philips, a global leader in health technology, announced it is working with leading medical centers ...


 December 31, 2020
December 31, 2020