June 2, 2026 — Results of an American College of Radiology-managed retrospective study involving 110,000 women presented at the 2026 ASCO Annual Meeting, shows that those who took GLP-1…
Breast Imaging
Women's health related to breast imaging, including mammography, breast MRI, ABUS, automated breast ultrasound, breast ultrasound, breast biopsy, PEM and positron emission mammography.
June 2, 2026 — Results of an American College of Radiology-managed retrospective study involving 110,000 women presented ...
May 6, 2026 — Artera, the developer of multimodal artificial intelligence (MMAI)-based prognostic and predictive cancer ...
April 16, 2026 – GE HealthCare has expanded its collaboration with DeepHealth, Inc., a wholly-owned subsidiary of RadNet ...
Despite decades of progress in breast imaging, one challenge continues to test even the most skilled radiologists ...
April 15, 2026 — QT Imaging Holdings, Inc. has launched its QTI Imaging-Olea Viewer, developed in collaboration with ...
April 1, 2026 — QT Imaging Holdings has released its latest image reconstruction software update, version 4.5.0. This ...
March 30, 2026 — Each year, the Alumni Association at the University of Missouri-Kansas City, recognizes the ...
While most women understand the importance of health screenings, an estimated 72 million have missed or postponed a ...
March 2, 2026 — A collaborative modeling study found that adding biennial breast magnetic resonance imaging (MRI) to ...
March 10, 2026 — QT Imaging Holdings has received U.S. Food and Drug Administration (FDA) 510(k) clearance for an ...
March 4, 2026 — QT Imaging Holdings recently announced that Mary W. Yamashita, MD will serve as medical advisor to the ...
The COVID-19 pandemic had a huge impact on the radiology community. Hospitals, doctors’ offices and clinics found ...
March 4, 2026 — Lunit has announced that 21 studies featuring its AI solutions will be presented at the European ...
Feb.23, 2026 — The first clinical patient received a Clairity Breast cancer risk score, marking a historic milestone in ...
Feb. 18, 2026 — Mammotome, a Danaher company, has introduced the Mammotome Prima MR Dual Vacuum-Assisted Breast Biopsy ...
Artificial Intelligence (AI) is becoming more common place in radiology practices, and emerging technologies are ...
Feb. 16, 2026 — Rising demand for breast cancer screening and diagnostics is outpacing the supply of available breast ...
Feb. 2, 2026 — Imagion Biosystems, Ltd. has submitted an Investigational New Drug (IND) application with the U.S. Food ...
Jan. 22, 2026 — In breast cancer, a biopsy is the only diagnostic procedure that can determine if a suspicious lump or ...
Jan. 27, 2026 — QT Imaging and Olea Medical have announced plans to collaborate. This collaboration enhances QT Imaging ...
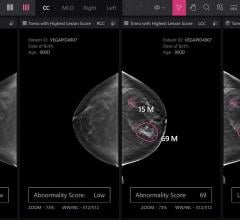
Jan. 16, 2026 — Vega Imaging Informatics has announced the successful curation of the world’s largest digital breast ...
Jan. 14, 2026 — VizMark has received U.S. Food and Drug Administration FDA 510k clearance for VM1, a non-metal visual ...
Jan. 14, 2026 — Susan G. Komen, the world’s leading breast cancer organization commends Assemblymember Lori Wilson (D ...
Jan. 14, 2026 — iSono Health has announced the commercial launch of ATUSA, an FDA-cleared, wearable and automated 3D ...







 June 02, 2026
June 02, 2026