May 29, 2026 — GE HealthCare recently announced that its MIM KineticID modeling software1 is now 510(k) pending with the U.S. Food and Drug Administration.
FDA
May 29, 2026 — GE HealthCare recently announced that its MIM KineticID modeling software1 is now 510(k) pending with the ...
May 27, 2026 — Subtle Medical has received FDA clearance for its SubtleHD (PET), the company's next-generation AI ...
May 19, 2026 — DeepHealth has received the CE Mark for the Brain Health and Brain Age solutions within its Neuro Suite ...
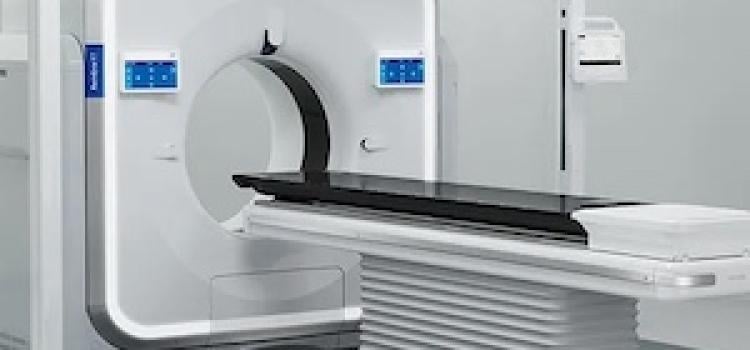
SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
May 21, 2026 — RADIN Health and AZmed have announced the expansion of their strategic partnership and enhance radiology ...
May 18, 2026 — ACIST Medical Systems, a Bracco company, has received U.S. Food and Drug Administration (FDA) clearance ...
May 12, 2026 — Siemens Healthineers has received clearance from the Food and Drug Administration for six new systems in ...
May 11, 2026 – At the International Society for Magnetic Resonance in Medicine (ISMRM) 2026 Annual Meeting, GE ...
May 6, 2026 — Artera, the developer of multimodal artificial intelligence (MMAI)-based prognostic and predictive cancer ...
April 23, 2026 — Royal Philips has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its ...
April 28, 2026 — Avatar Medical has been granted FDA 510(k) clearance for Avatar Medical Vision, its software platform ...
April 28, 2026 — Abbott has received U.S. Food and Drug Administration (FDA) clearance and CE Mark for its next ...
April 23, 2026 — On April 23, GE HealthCare announced the first patient has been dosed in the international, multi ...
April 16, 2026 — Royal Philips has received U.S. Food and Drug Administration 510(k) clearance for the Philips Spectral ...
April 2, 2026 — GE HealthCare has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for True ...
March 30, 2026 — Butterfly Network, Inc. has received clearance from the U.S. Food and Drug Administration (FDA) for a ...
March 24, 2026 — MARS Bioimaging, a New Zealand–headquartered medical device company, has received U.S. Food and Drug ...
March 20, 2026 — Siemens Healthineers recently announced its Varian TrueBeam radiotherapy systems, which include ...
March 18, 2026 — FluoGuide A/S, a biotech company maximizing surgical outcomes in oncology, has announced that the U.S ...
March 10, 2026 — QT Imaging Holdings has received U.S. Food and Drug Administration (FDA) 510(k) clearance for an ...
Mar. 9, 2026 — GE HealthCare recently announced that View, the viewer within the Genesis Radiology Workspace, has ...


 May 29, 2026
May 29, 2026 



















