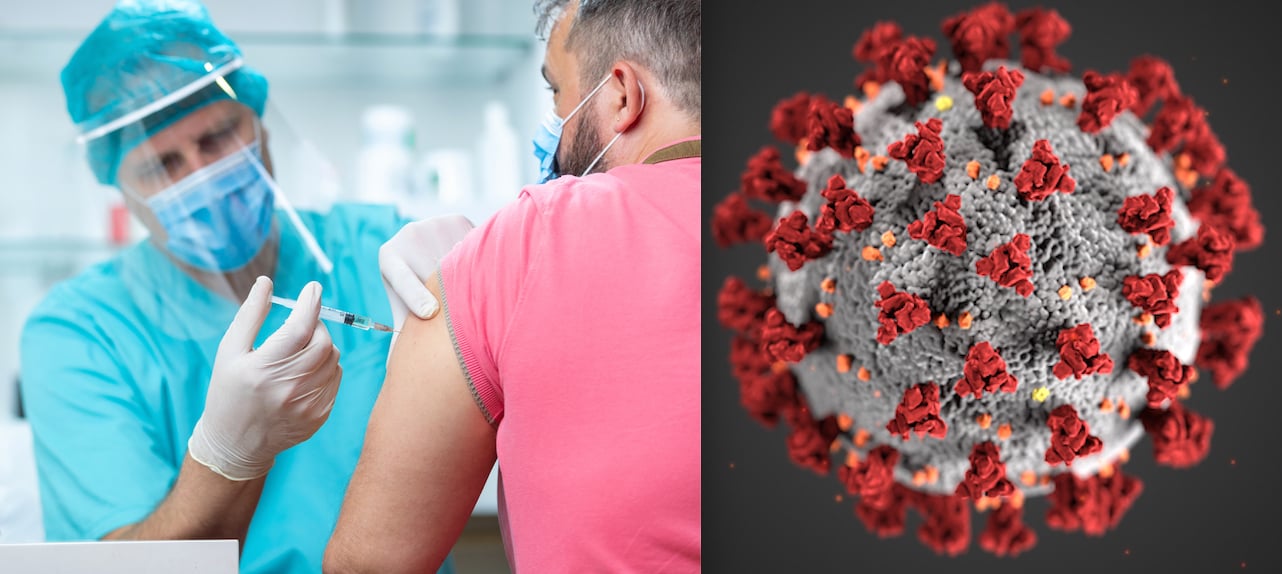
December 14, 2020 — The U.S. Food and Drug Administration (FDA) issued the first emergency use authorization (EUA) for the COVID-19 vaccine submitted by Pfizer Inc. in partnership with BioNTech Manufacturing GmbH. It is the first vaccine to gain an FDA clearance for use and will likely be followed by an approval for a second vaccine by Moderna very soon.
The indication for the vaccine is for the prevention of coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in individuals 16 years of age and older.
The Pfizer-BioNTech COVID-19 Vaccine (BNT162b2) is a suspension for intramuscular injection administered as a series of two doses (0.3 mL each) 3 weeks apart.
The EUA cleared the way for shipments of the vaccine to start rolling out Sunday to about 145 larger hospitals equipped to store and deliver the vaccine, which must be stored in special freezers at 70 degrees below zero. Vaccinations for front-line hospital staff began Monday, Dec. 14, and the plan is to vaccinate close to 20 million people with the first dose by the end of December. Those receiving the vaccine need a second booster injection 21 days later.
“It is nothing short of a medical miracle to have FDA authorization of a vaccine for COVID-19 just over 11 months since the virus was made known to the world," said HHS Secretary Alex Azar. "This vaccine, like any vaccine FDA potentially authorizes, has been through multiple stages of safety review, and it has shown extra-ordinary effectiveness in protecting people from the virus. Vaccines will help bring this pandemic to an end, which is all the more reason to double down on the public health measures we need to stay safe in the coming months."
The FDA's Vaccines and Related Biologics Advisory Committee met Friday, Dec. 10 to discuss the effectiveness data for the Pfizer vaccine and recommended that the agency grant emergency authorization. Final approval came from the FDA Saturday.
Following the FDA decision, Operation Warp Speed (OWS) and private sector partners executed their plan for distributing the first allocation of doses to sites designated by the public health jurisdictions and five federal agencies with which OWS and CDC have been working to plan distribution.
Emergency Use Authorization for Vaccines Explained
Storage and Handling and the Pfizer mRNA COVID-19 Vaccine
The vaccine uses messenger RNA, which is extremely fragile and requires very cold storage temperatures and can be damaged by light. This is the is the first ever messenger RNA vaccine to gain FDA clearance.
Cartons of Pfizer-BioNTech COVID-19 Vaccine Multiple Dose Vials are shipped in thermal containers with dry ice. Once received, hospitals need to remove the vial cartons immediately from the thermal container and store in an ultra-low temperature freezer between -112ºF to -76ºF (-80ºC to -60ºC). Vials must be kept frozen and have minimized exposure to room light, and avoid exposure to direct sunlight and ultraviolet light.
If an ultra-low temperature freezer is not available, the thermal container in which the Pfizer-BioNTech COVID-19 Vaccine arrives may be used as temporary storage when consistently re-filled to the top of the container with dry ice.
For use, the vaccine must be thawed and then can be stored in the refrigerator between 35ºF to 46ºF (2ºC to 8ºC) for up to 5 days. A carton of 25 vials or 195 vials may take up to 2 or 3 hours to thaw in the refrigerator.
For immediate use, undiluted vials can be thawed at room temperature for 30 minutes. Thawed vials can be handled in room light conditions. Vials must reach room temperature before dilution.
After dilution, the vaccine must be used within 6 hours from the time of dilution. After 6 hours the vaccine needs to be discarded and cannot be refrozen.
Read more on the FDA instruictions for the use and handling of the Pfizer-BioNTech COVID-19 Vaccine.
Limitations and Safety Concerns of the Pfizer Vaccine
The FDA's Advisory Committee on Immunization Practices (ACIP) panelists met Dec. 11 and recommended approval of the vaccine for those age 16 and older. The panel said clinicians need more guidance on whether to use the vaccine on pregnant or breastfeeding women, the immunocompromised, those who have a history of allergies and those under age 16.
The FDA-issued healthcare provider information sheet on the vaccine said there is not enough data to recommend vaccinating those women or the immunocompromised, and also advises against giving the vaccine to individuals who have a history of serious allergic reaction to any component of the vaccine.
While the vaccine appears to be very safe, these and other questions remain because of the rush to get the vaccine into use and limited trial data for these types of patients.
Side Effects of the Pfizer COVID-19 Vaccine
Pfizer says on its website for the vaccine that in clinical studies, adverse reactions in participants 16 years of age and older included:
• Pain at the injection site (84.1%)
• Fatigue (62.9%)
• Headache (55.1%)
• Muscle pain (38.3%)
• Chills (31.9%)
• Joint pain (23.6%)
• Fever (14.2%)
• Injection site swelling (10.5%)
• Injection site redness (9.5%)
• Nausea (1.1%)
• Malaise (0.5%)
• Lymphadenopathy (0.3%)
Pfizer also said severe allergic reactions have been reported following the Pfizer-BioNTech COVID-19 Vaccine during mass vaccination outside of clinical trials. The company also warned additional adverse reactions, some of which may be serious, may become apparent with more widespread use of the Pfizer-BioNTech COVID-19 Vaccine.
Immunocompromised people, including individuals receiving immunosuppressant therapy, may have a diminished immune response to the Pfizer-BioNTech COVID-19 Vaccine, according to Pfizer.
Moderna COVID Vaccine May See FDA Clearance in Next Week
Moderna's mRNA-1273 COVID vaccine also has been submitted for FDA EUA clearance, which is widely expected to gain approval before Christmas.
A positive sign of this is the Dec. 11 announcement by the U.S. Department of Health and Human Services (HHS) that it is purchasing 100 million doses of the Moderna vaccine.
Under the agreement, Moderna will leverage its U.S.-based manufacturing capacity to fill, finish and ship vials of mRNA-1273 as the bulk material is produced. The additional doses ordered today provide for continuous delivery through the end of June 2021. This strategy will help meet the anticipated demand for mRNA-1273 and safely accelerate the delivery schedule for all 200 million doses the U.S. government is purchasing.
The purchase brings the total doses of mRNA-1273 owned by the federal government to 200 million. Moderna began manufacturing the first 100 million doses while clinical trials were getting underway. Manufacturing in parallel with clinical trials expedited the traditional vaccine development timeline and built toward the U.S. government’s Operation Warp Speed goal to begin delivering safe and effective vaccines to the American people by the end of the year.
The Biomedical Advanced Research and Development Authority (BARDA), part of the HHS Office of the Assistant Secretary for Preparedness and Response, has now collaborated with the DoD Joint Program Executive Office for Chemical, Biological, Radiological and Nuclear Defense and Army Contracting Command to provide up to approximately $3.2 billion to expand the manufacturing capacity reserved for mRNA-1273 and to deliver the vaccine doses to government-designated locations across the country. This federal funding brings the total provided to Moderna for this vaccine, including vaccine development, clinical trials and manufacturing, to $4.1 billion. The government also has the option to acquire up to an additional 300 million doses of the Moderna vaccine.
The vaccine candidate was co-developed by Moderna and scientists from the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, with NIAID also supporting the vaccine’s nonclinical studies and clinical trials. BARDA supported phase 2/3 clinical trials, vaccine manufacturing scale up and other development activities for this vaccine.
The Phase 3 clinical trial, which began July 27, was the first government-funded Phase 3 clinical trial for a COVID-19 vaccine in the U.S. and enrolled approximately 30,000 adult volunteers who did not have COVID-19. An independent data safety monitoring board overseeing the Phase 3 clinical trial reviewed the trial data and concluded that the vaccine was safe, prevented disease in 94 percent of the volunteers who received the vaccine and was generally well tolerated. The clinical studies are ongoing.
Moderna is the second company to apply for FDA emergency use authorization of a coronavirus vaccine that uses messenger RNA (mRNA), and FDA has scheduled a Vaccines and Related Biological Products Advisory Committee meeting Dec. 17 to review the company’s request.
Under Operation Warp Speed, federal partners are working with multiple companies – Moderna, Pfizer, Janssen (Johnson&Johnson), AstraZeneca, Novavax, and Sanofi in collaboration with GSK – to develop vaccines that use a variety of vaccine platform technologies and manufacture the COVID-19 vaccine doses while clinical trials are underway. If any of these vaccine candidates are authorized by the U.S. Food and Drug Administration for emergency use, as outlined in agency guidance, HHS and DOD can negotiate agreements with the respective companies to purchase enough vaccine to meet demand in the United States.
COVID-19 Vaccines Are the First to Use Messenger RNA
The Pfizer and Moderna vaccines are the first vaccines being commercialized that use messenger RNA (mRNA). This has been discussed for years as a novel way to speed development of new vaccines and greatly simplify vaccine production. Messenger RNA vaccines take advantage of the process that cells use to make proteins, or a piece of a protein, in order to trigger an immune response and build immunity to a virus.
In contrast, most vaccines today use weakened or inactivated versions or components of a disease-causing virus to stimulate the body’s immune response to create antibodies.
Since mRNA vaccines do not use the live virus that causes COVID-19, the vaccine cannot give recipients the virus.
COVID-19 mRNA vaccines give instructions for our cells to make a harmless piece of what is called the “spike protein.” The spike protein is found on the surface of the virus that causes COVID-19.
COVID-19 mRNA vaccines are given in the upper arm muscle. Once the instructions (mRNA) are inside the muscle cells, the cells use them to make the protein peice. After the protein piece is made, the cell breaks down the instructions and gets rid of them.
Next, the cell displays the protein piece on its surface. Our immune systems recognize that the protein doesn’t belong there and begin building an immune response and making antibodies, like what happens in natural infection against COVID-19.
At the end of the process, the body learned how to protect against future infection. The benefit of mRNA vaccines is those vaccinated gain this protection without ever having to risk the serious consequences of getting sick with COVID-19.
Prior to the FDA EUA Saturday, the CDC said there are currently no licensed mRNA vaccines in the United States. However, researchers have been studying and working with them for decades. Interest has grown in these vaccines because they can be developed in a laboratory using readily available materials. This means the process can be standardized and scaled up, making vaccine development faster than traditional methods of making vaccines.
mRNA vaccines have been studied before for flu, Zika, rabies, and cytomegalovirus (CMV). As soon as the necessary information about the virus that causes COVID-19 was available, scientists began designing the mRNA instructions for cells to build the unique spike protein into an mRNA vaccine.
The CDC said future mRNA vaccine technology may allow for one vaccine to provide protection for multiple diseases, thus decreasing the number of shots needed for protection against common vaccine-preventable diseases.
Vaccine Development 101 Explained by the FDA
FDA Issues Alert Regarding SARS-CoV-2 Viral Mutation to Healthcare Providers — Updated January 2021
Related COVID Radiology Content:
Lung Ultrasounds Could Help Determine COVID-19 Outcome
Johns Hopkins Medicine Expert Weighs Devastating Impact of COVID-19 on Healthcare Workers
CT in a Box Helps Rapidly Boost Imaging Capability at COVID Surge Hospitals
CT Imaging of the 2019 Novel Coronavirus (2019-nCoV) Pneumonia
Infervision in the Frontlines Against the Coronavirus
CT Imaging Features of 2019 Novel Coronavirus (2019-nCoV)
The Cardiac Implications of Coronavirus
VIDEO: COVID-19 Pneumonia Chest CT Scan Scroll Through
CT Imaging of the 2019 Novel Coronavirus (2019-nCoV) Pneumonia
Chest CT Can Distinguish Negative From Positive Lab Results for COVID-19
VIDEO: CT Sees Increased Use During COVID-19
Radiologists Urge Use of Medical Imaging and AI-Powered Solutions to Manage COVID-19
Stroke Scans Could Reveal COVID-19 Infection
Handheld Ultrasound Used to Monitor COVID-19 Patients With Cardiac Complications
Study Looks at CT Findings of COVID-19 Through Recovery
Using Lung X-rays to Diagnose COVID-19
How COVID-19 Affects the Brain in Neuroimaging
VIDEO: Lingering Myocardial Involvement After COVID-19 Infection — Interview with Aaron Baggish, M.D.
COVID-19 Can Impact Hearts in Young Children
VIDEO: What Are The Long-term Cardiac Impacts of COVID-19 Infection — Interview with Todd Hurst, M.D.


 July 25, 2025
July 25, 2025