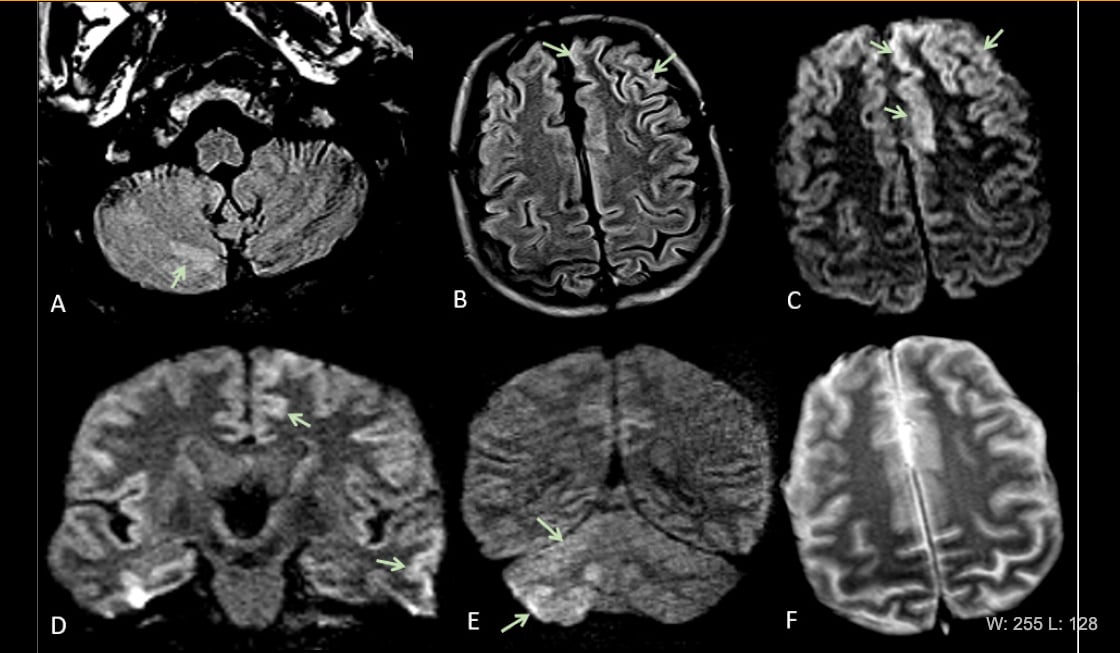
This is Figure 2 from the article in Radiology: Acute encephalopathy. A 60 year-old-man without history of seizures presenting with convulsion. (A-B) Multifocal areas of FLAIR hyperintensity in the right cerebellum (arrows in A), left anterior cingular cortex and superior frontal gyrus (arrows in B). (C-D) Restricted diffusion in the left anterior cingulate cortex, superior frontal and middle temporal gyrus (arrows in D) and right cerebellum (arrows in E), consistent with cerebellar diaschisis. F) No hemosiderin deposits in gradient echo sequences.
Four recent radiology studies, from New York, Italy, Iran and China, show how COVID-19 (SARS-CoV-2) impacts the brain of some infected patients. Accumulating evidence suggests that a subgroup of patients with severe COVID-19 might have a cytokine storm syndrome which could be a trigger for ischemic strokes. Other abnormalities have also been found in COVID-19 patient brain imaging studies.
COVID-19 was originally thought to resemble the flu, with pneumonia lung manifests. However, it has become clear the past couple months that the virus also attacks the brain, heart, kidneys, vascular system, caused severe inflammatory responses, causes venous thromboembolism (VTE) and can cause multi-organ failure. Within the brain, COVID-19 patients have been observed with ischemic stroke, intracranial hemorrhage, focal cerebral arteriopathy, hypoxic-ischemic encephalopathy, encephalitis and acute hemorrhagic necrotizing encephalopathy.
“Currently, we have a poor mechanistic understanding of the neurological symptoms in COVID-19 patients, whether these are arising from critical illness or from direct central nervous system invasion of SARS-CoV-2,” wrote the authors of the first study, "Imaging in Neurological Disease of Hospitalized COVID-19 Patients: An Italian Multicenter Retrospective Observational Study," which was published as a research letter in the journal Radiology.[1]
The study sought to systematically characterize neurological symptoms and neuroimaging features in hospitalized COVID-19 patients from multiple institutions in Italy. That country was the second epicenter of the spread of the disease when it left China in February, and at the time of study writing, Italy had 222,104 confirmed COVID-19 cases and 31,106 deaths. Several studies have described the spectrum of chest imaging features of COVID-19,[2] However, to date, only a few case reports have described COVID-19 associated neuroimaging findings.[3–8] This study tried to consolidate the neuro-imaging COVID-19 findings into one document.
Of 725 consecutive hospitalized patients with COVID-19, 108 (15%) had acute neurological symptoms requiring neuroimaging. It is the largest study to date of neuroimaging findings in patients with COVID-19 hospitalized in multiple institutions in Italy. The most common neurological symptoms were altered mental status in 64 (59%) patients and ischemic stroke in 34 (31%) patients. Find a complete list of characteristics for the COVID-19 patients in Figure 1 of this article.
Of the 108 patients with ischemic stroke, 31 (29%) had no known past medical history and 77 (71%) had at least one of the following chronic disorders: coronary artery disease 25 (23%), cerebrovascular disease 15 (14%), hypertension 55 (51%) and diabetes 30 (28%). Of the 31 (29%) patients with no known past medical history (age range 16-62 years), 10 had acute ischemic infarcts and 2 had intracranial hemorrhage.
All imaging was obtained using magnetic resonance imaging (MRI) brain and spine scans, performed on 1.5T scanners with standardized protocols. For contrast studies, Gd-DTPA (0.1 mmol/kg gadobutrol [Gadovist]) was used.
Scans showed 47% of patients (51 out of 108) had acute neuroimaging abnormalities. These included:
• 34 (31% with acute ischemic infarcts
• 6 (6%) intracranial hemorrhage
• 2 (12%) cerebral venous thrombosis
• 2 (10%) multiple sclerosis (MS) plaque exacerbation
• 2 (10%) nonspecific encephalopathy
• 2 (10%) Guillian-Barre syndrome (rare disorder in which the body's immune system attacks the nerves)
• 1 (10%) Miller-Fisher syndrome (rare, acquired nerve disease with abnormal muscle coordination, paralysis of the eye muscles and absence of the tendon reflexes. Usually preceded by a viral illness.)
• 1 (5%) posterior reversible encephalopathy syndrome (PRES). Characterized by a headache, seizures, altered mental status and visual loss, and by white matter vasogenic edema.
• 1 (5%) acute encephalopathy
"Our study demonstrated that the neuroimaging features of hospitalized COVID-19 patients were variable, without specific pattern, but dominated by acute ischemic infarcts and intracranial hemorrhages. We also showed that MR neuroimaging spectrum may include posterior reversible encephalopathy syndrome (PRES), hypoxic-ischemic encephalopathy, exacerbation of demyelinating disease and nonspecific cortical pattern of T2 FLAIR hyperintense signal with associated restriction diffusion that may be caused by systemic toxemia, viremia and/or hypoxic effects," the authors concluded.
The authors also noted a lower prevalence of central nervous system (CNS) symptoms than the Wuhan, China, experience [8] (15 vs. 25%). However the prevalence of ischemic strokes was higher in the Italian study (31 vs. 11%).
COVID-19 Can Cause Diffuse Disorders of White Matter and Micro-hemorrhages
Diffuse leukoencephalopathy and juxtacortical/callosal microhemorrhages were brain imaging features in critically ill COVID-19 patients found in a study from the New York University School of Medicine.[9] The study reported brain imaging features in 11 critically ill COVID-19 patients with persistently depressed mental status who underwent MRI between April 5-25, 2020.
These features included:
• Confluent T2 hyperintensity and mild restricted diffusion in bilateral supratentorial deep and subcortical white matter (in 10 of 11 patients).
• Multiple punctate microhemorrhages in juxtacortical and callosal white matter (in 7 of 11 patients).
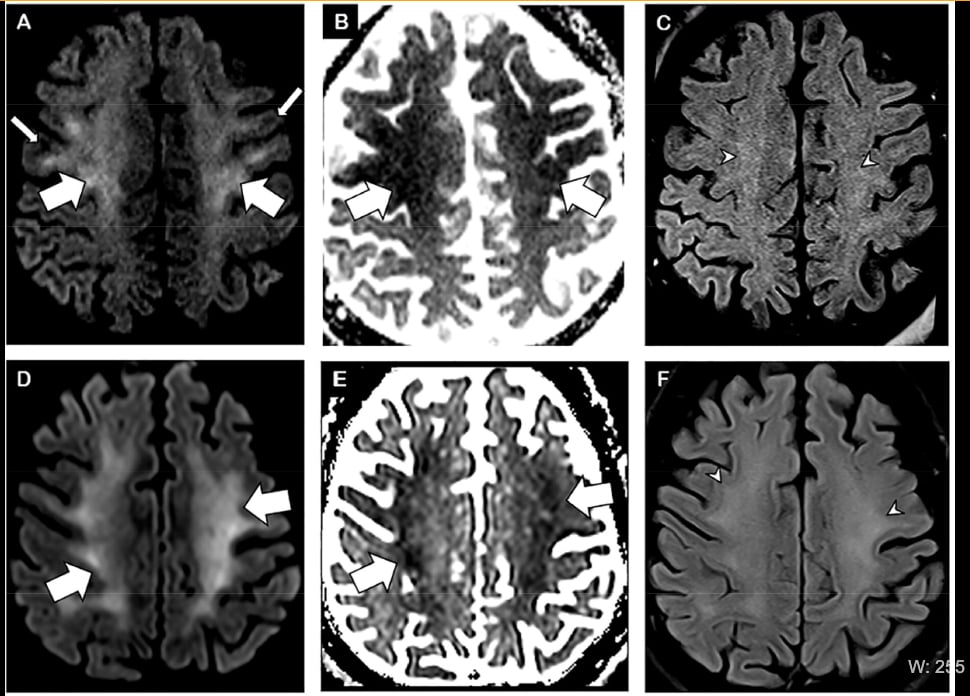
Figure 1 from the Radiology article. Brain MRI in two critically ill COVID-19 patients with persistently depressed mental status including a 56-year old man (A-C), and a 64-year old man (D-F). Axial diffusion-weighted (A, D), apparent diffusion coefficient (B, E) and FLAIR (C, F) images at the level of centrum semiovale in both patients demonstrate symmetric diffuse T2/FLAIR hyperintensity (arrowheads) and mild restricted diffusion (thick arrows) involving the deep and subcortical white matter with relative sparing of juxtacortical white matter (thin arrows) in both patients. The restricted diffusion is more conspicuous than the T2/FLAIR hyperintensity.
Researchers said abnormalities extended from the precentral gyrus down to the centrum semiovale and corona radiata. At the level of temporal and occipital horns, the posterior cerebral white matter in all patients was involved more than the anterior. The deep gray nuclei were spared. The juxtacortical white matter was also relatively spared, with the exception of the pre-central gyrus in all 10 patients and occipital lobes in seven patients. One patient received intravenous contrast for the MRI and there was no abnormal intracranial enhancement.
The infratentorial parenchyma tended to be less affected, seen only in four patients with mild involvement of middle cerebellar peduncles and medial cerebellar hemispheres.
The study includes numerous images showing these findings.
COVID-19 Pediatric Patient With Focal Cerebral Arteriopathy
In research published in Radiology on June 2, researchers in Iran and Children’s Hospital of Philadelphia presented a case of focal cerebral arteriopathy (FCA) and ischemic stroke in a pediatric COVID-19 patient who presented with seizure, right hemiparesis (weakness), and dysarthria (difficulty speaking) and who tested positive for SARS-CoV2 virus both from nasopharyngeal swab and cerebral spinal fluid.[10] FCA is a common cause of ischemic stroke in previously healthy children.
The researchers said the neuroinvasive mechanism of SARS-CoV-2 is not fully understood. It may enter the CNS through the blood, with other proposed routes, including the olfactory and trigeminal nerves, cerebral spinal fluid (CSF) and the lymphatic system.[11] Neurologic manifestations in COVID-19 pediatric patients are not common, but are reported, and this is this one such presumptive case of ischemic stroke due to FCA associated with COVID-19.
The case involves a previously healthy 12-year-old-boy with new onset of generalized seizures, initially treated with Diazepam. Shortly after, he developed right-sided hemiparesis and dysarthria. There was no previous history of fever, cough, shortness of breath, skin rash, hemoglobinopathy or recent trauma. No one in his family had a known history of COVID-19 infection.
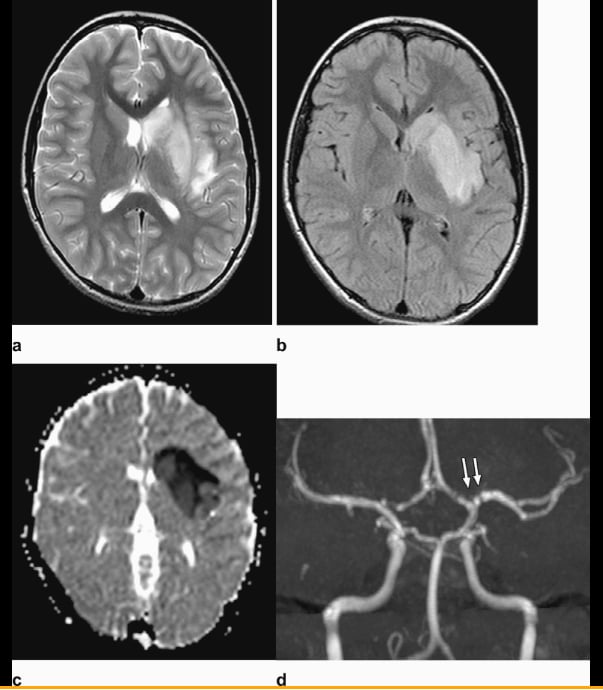
Figure 2 from the Radiology article: Axial T2-weighted (a) and FLAIR (b) Magnetic resonance imaging show diffuse hyperintense signal and edema of the caudate nucleus head, putamen, anterior limb of the internal capsule, and parts of external capsule and insula on the left side, with corresponding low values on the axial apparent diffusion coefficient map, in keeping with an acute infarct. Time-of-flight magnetic resonance angiography maximal intensity projection reformatted image demonstrates focal irregular narrowing and banding of the proximal left M1 segment of the middle cerebral artery with a slightly reduced distal flow in the middle cerebral artery.
There were no symptoms of other systems involvement, cervical enlarged lymph nodes, rash or redness. The patient had no respiratory abnormalities. The patient received conservative management. The patient is at home undergoing rehabilitation.
The researchers said the major imaging findings in this patient are the presence of unilateral focal vasculopathy, characterized by focal irregular narrowing and banding of the proximal left middle cerebral artery associated with left basal ganglia and insula acute infarct. There are many potential differential considerations in a child with acute stroke, including FCA, arterial dissection, embolic or thrombotic stroke, cerebral vasculitis in the context of systemic disease (including Kawasaki disease), and genetic disorders with arteriopathy, among others. Their leading diagnosis is FCA, which is defined by Wintermark et al.[13] Based on the patient’s history and lack of thrombotic processes outside the CNS, embolic stroke and dissection were considered less likely than FCA.
There have been numerous reports of multi-system inflammatory syndrome in children (MIS-C), which causes an elevated immune response in COVID-19 pediatric patients that process Kawasaki disease-like presentations. There was no evidence to suggest a multi-system inflammatory syndrome or Kawasaki-like presentation, as the patient did not show any of the classical clinical physical findings, the researchers said.
Read more in the article Kawasaki-like Inflammatory Disease Affects Children With COVID-19.
Encephalitis as a Clinical Manifestation of COVID-19
Viral infections have detrimental impacts on neurological functions and can cause severe neurological damage. A study from China showed coronavirus can exhibit neurotropic properties and may also cause neurological diseases. Researchers looked at neurological complications in COVID infections and the possible mechanisms of damage to the nervous system.[13] They found most neurological diseases could be caused by coronavirus invasion and COVID-19 causes nerve damage via diverse pathways.
On March 4, 2020, Beijing Ditan Hospital reported the first case of viral encephalitis caused by novel coronavirus attacking the central nervous system. The researchers confirmed the presence of SARS-CoV-2 in the cerebrospinal fluid by genome sequencing. It illustrated that COVID-19 has potential to cause nervous system damage.
A Wuhan male had fever, shortness of breath and myalgia since Jan 28. On Feb 10, his disease deteriorated and his consciousness suddenly progressed to confusion. Laboratory tests indicated low WBC count (3.3 x 109/L) and lymphopenia (0.8 x 109/L). Skull computed tomography (CT) was normal, while chest CT showed multiple subpleural ground glass opacities (GGOs). The patient was positive for SARS-CoV-2. On physical examination, vital signs were stable without altered consciousness. Meningeal irritation signs (including nuchal rigidity, Kernig sign and Brudzinski sign) and extensor plantar response were present.
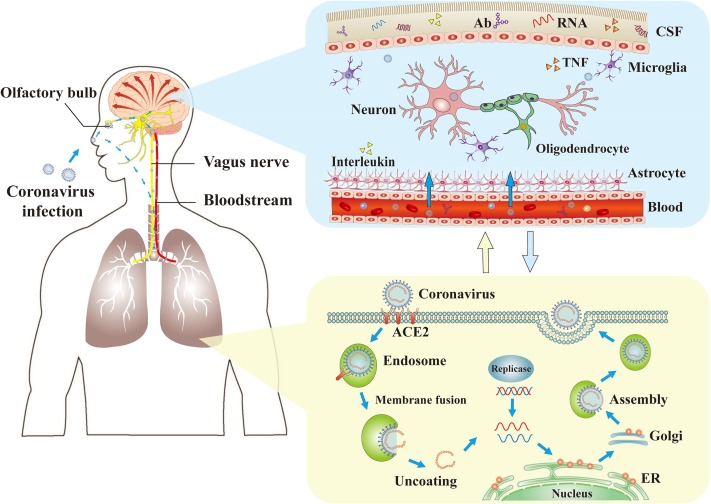
Figure 1 from the journal Brain, Behavior, and Immunity. The mechanisms of coronaviruses infections and neurological damage caused by coronaviruses. The coronaviruses can cause nerve damage through direct infection pathways (blood circulation pathways and neuronal pathways), hypoxia, immune injury, ACE2 and other mechanisms. Meanwhile, the coronaviruses have detrimental effects to attack the lung tissue, and causes a series of lung lesions such as hypoxia. Furthermore, the coronaviruses can enter the nervous system directly through the olfactory nerve, and also enter the nervous system through blood circulation and neuronal pathways, resulting in neurological disorders. Ab: antibody; ACE2: angiotensin-converting enzyme 2; CSF: cerebrospinal fluid; ER: endoplasmic reticulum; TNF: tumor necrosis factor.
The study said arterial blood gas analysis indicted PO2/FiO2 ratio of 240 mmHg. This COVID-19 patient was treated with arbidol and oxygen therapy, whereas his consciousness was not improved. Lumbar puncture was performed on Feb. 16, and the cerebrospinal fluid (CSF) pressure was 220 mmHg. Laboratory tests with CSF showed WBC (0.001 x 109/L), protein (0.27 g/L), ADA (0.17 U/L) and sugar (3.14 mmol/L) contents within normal limits. The CSF specimen was further tested for SARS-CoV-2 but the result was negative.
This patient did not have evidence of bacterial or tuberculous infection of the CNS. After careful evaluation by neurologic experts, encephalitis associated with COVID-19 infection was concluded.
Treatment was largely supportive, including mannitol infusion. The patient’s consciousness was generally unsatisfactory since Feb. 20, and chest CT suggested a resolution of GGOs. The patient regained clear consciousness Feb. 24. Two consecutive throat swab tests were negative for SARS-CoV-2. The patient was discharged from hospital Feb. 27.
All RSNA COVID-19 research articles can be found at Special Focus: COVID-19. Additional COVID-19 resources can be found at RSNA.org/COVID-19.
References:
3. Li Y, Wang M, Zhou Y, Chang J, Xian Y, Mao L et al. Acute Cerebrovascular Disease Following COVID-19: A Single Center, Retrospective, Observational Study. SSRN Electronic Journal 2020;10.2139/ssrn.3550025.


 May 05, 2026
May 05, 2026