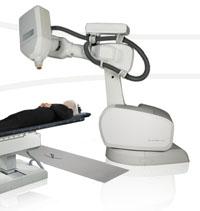
August 30, 2011 — Accuray Inc. announced a significant increase in use of the CyberKnife Robotic Radiosurgery System for prostate SBRT (stereotactic body radiotherapy). In Q4 2011, the company experienced a 24 percent increase in worldwide prostate treatments over Q4 2010 and a 17 percent increase over Q3 2011.
Earlier in 2011, Accuray announced the first publication from the expanding foundation of maturing prostate SBRT long-term studies documenting five-year outcomes of prostate cancer patients treated with the CyberKnife system. The multi-center study combines data from patients at Stanford University in Stanford, Calif. and Naples Community Hospital in Naples, Fla. It found 93 percent of patients had no recurrence of their cancer at a median follow-up of five years. That rate compares favorably to results obtained with other treatment modalities, including surgery and conventional radiation therapy.
The study also found generally low levels of urinary and rectal toxicity, concluding that CyberKnife SBRT can achieve high rates of disease control while sparing critical structures; this minimizes undesirable side effects typically associated with other prostate cancer treatments and preserving patients' quality of life.
Adding to the growing body of evidence is the completion of patient accrual in an Accuray-sponsored, multi-center homogeneous prostate study for low and intermediate risk prostate cancer. The study consists of teams at the Swedish Cancer Center in Seattle and the Beth Israel Deaconess Medical Center in Boston and involves 21 centers in the United States. A total of 309 eligible patients have been accrued and are being followed closely to allow precise evaluation of the long-term efficacy and toxicity outcomes following CyberKnife prostate SBRT.
Michael Kendrick, M.D., a practicing physician familiar with radiosurgery, was the first patient in the world to undergo CyberKnife prostate SBRT and was treated in 2003 at Stanford.
"After learning about the CyberKnife and drawing on my expertise as a physician, deciding to move forward with CyberKnife prostate SBRT was a no brainer," said Kendrick. "CyberKnife's pinpoint accuracy and ability to track and correct for prostate motion was an important factor in my decision. I experienced no side effects, a fast recovery time, and was able to maintain a high quality of life during and following treatment. My PSA level has remained stable proving to me that CyberKnife SBRT was an effective treatment, a choice I have never regretted."
The system is a non-invasive treatment option for prostate cancer with the ability to deliver targeted and destructive doses of radiation at high accuracy, enabling treatment of tumors close to critical, healthy structures. Its unique ability to not only track but automatically correct for unpredictable movement of the prostate during treatment minimizes radiation exposure to health tissues; it also helps patients avoid many side effects typically associated with other treatment options, preserving quality of life.
Accuray continues to support patient education, including awareness of all treatment options. The company also supports access to innovation and high quality care through partnerships with leading non-profit prostate cancer organizations, including:
- Us TOO International Prostate Cancer Education & Support Network
Founded in 1990 by five men treated for prostate cancer, Us TOO is a grassroots, registered 501(c)(3) non-profit prostate cancer education and support network of 325 support group chapters worldwide. The organization's mission is to provide men and their families with free information, materials and peer-to-peer support to make informed choices on detection, treatment options and coping with ongoing survivorship. Accuray recently collaborated with Us TOO on a new, comprehensive information guide addressing what every newly diagnosed prostate cancer patient needs to know about treatment with the CyberKnife system.
- ZERO — The Project to End Prostate Cancer
ZERO is committed not only to reducing prostate cancer and alleviating pain from the disease, but to ending it altogether. The organization provides comprehensive treatment information to patients, education to those at risk and free prostate cancer testing. In addition, ZERO works to increase funds from the federal government for research into new treatments. It also provides local grants to end prostate cancer through its national race and event series, the Great Prostate Cancer Challenge.
For more information: www.accuray.com


 May 06, 2026
May 06, 2026 









