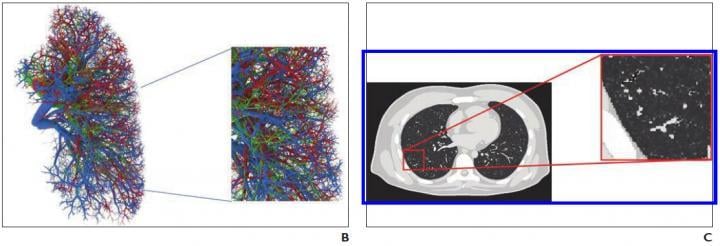
B, Representative computational model shows lung stroma intraorgan structure of XCAT phantom that was developed using anatomically informed mathematic model. Inset shows enlarged view for better visibility of details and small structures. C, Voxelized rendition (ground truth) of XCAT phantom highlights detailed model of lung parenchyma. Inset shows enlarged view for better visibility of details and small structures. Image courtesy of the American Roentgen Ray Society (ARRS), American Journal of Roentgenology (AJR)
August 26, 2020 — An open-access article in ARRS' American Journal of Roentgenology (AJR) established a foundation for the use of virtual imaging trials in effective assessment and optimization of computed tomography (CT) and radiography acquisitions and analysis tools to help manage the coronavirus disease (COVID-19) pandemic.
Virtual imaging trials have two main components — representative models of targeted subjects and realistic models of imaging scanners — and the authors of this AJR article developed the first computational models of patients with COVID-19, while showing, as proof of principle, how they can be combined with imaging simulators for COVID-19 imaging studies.
"For the body habitus of the models," lead author Ehsan Abadi explained, "we used the 4-D extended cardiac-torso (XCAT) model that was developed at Duke University."
Abadi and his Duke colleagues then segmented the morphologic features of COVID-19 abnormalities from 20 CT images of patients with multidiagnostic confirmation of SARS-CoV-2 infection and incorporated them into XCAT models.
"Within a given disease area, the texture and material of the lung parenchyma in the XCAT were modified to match the properties observed in the clinical images," Abadi et al. continued.
Using a specific CT scanner (Definition Flash, Siemens Healthineers) and validated radiography simulator (DukeSim) to help illustrate utility, the team virtually imaged three developed COVID-19 computational phantoms.
"Subjectively," the authors concluded, "the simulated abnormalities were realistic in terms of shape and texture," adding their preliminary results showed that the contrast-to-noise ratios in the abnormal regions were 1.6, 3.0, and 3.6 for 5-, 25-, and 50-mAs images, respectively.
For more information: www.arrs.org
Related Coronavirus Content:
VIDEO: Imaging COVID-19 With Point-of-Care Ultrasound (POCUS)
Cardiac Imaging Best Practices During the COVID-19 Pandemic
RSNA Publishes COVID-19 Best Practices for Radiology Departments
ASE Guidelines for the Protection of Echocardiography Providers During the COVID-19 Outbreak
New CT Scoring Criteria for Timely Diagnosis, Treatment of Coronavirus Disease
FDA Issues New Policy for Imaging Systems During COVID-19
VIDEO: COVID-19 Precautions for Cardiac Imaging — Interview with Stephen Bloom, M.D.
A Review of Studies Cautions Against Chest CT for Coronavirus Diagnosis
New Research Finds Chest X-ray Not Reliable Diagnostic Tool for COVID-19
VIDEO: Radiology Industry Responding to COVID-19
University of Washington Issues Radiology Policies for COVID-19
VIDEO: Best Practices for Nuclear Cardiology During the COVID-19 Pandemic — Interview with Hicham Skali, M.D.
New Research Highlights Blood Clot Dangers of COVID-19
Survey Reveals Most Medical Practices are Now Using Telehealth Due to COVID-19
CMS Offers Recommendations on Reopening Healthcare in Areas of Low COVID-19 Cases
CT Provides Best Diagnosis for Novel Coronavirus (COVID-19)
Radiology Lessons for Coronavirus From the SARS and MERS Epidemics
Radiologists Describe Coronavirus CT Imaging Features
CT Imaging of the 2019 Novel Coronavirus (2019-nCoV) Pneumonia
ACC COVID-19 recommendations for the cardiovascular care team
VIDEO: What Cardiologists Need to Know about COVID-19 — Interview with Thomas Maddox, M.D.


 April 29, 2026
April 29, 2026 









