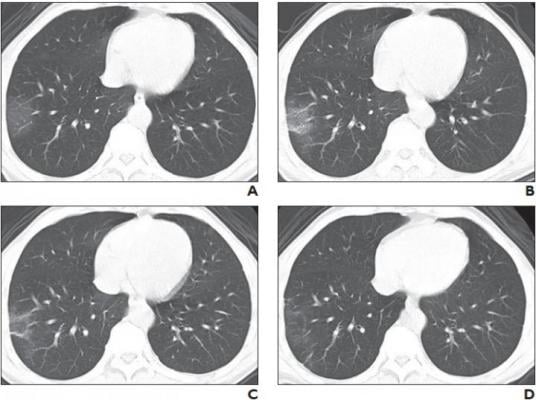
CT scoring criteria were applied to images from sequential chest CT examinations. A, Initial chest CT image obtained 2 days after onset of symptoms shows small region of subpleural ground-glass opacities in right lower lobe, for CT score of 1. B, Chest CT image obtained on day 3 of treatment shows slightly enlarged region of subpleural ground-glass opacities with partial crazy-paving pattern and consolidation, for CT score of 3. C, Chest CT image obtained on day 5 of treatment shows partial resolution of consolidation, for CT score of 2. D, Chest CT image obtained on day 14 of treatment shows continued resolution of consolidation with minimal residual ground-glass opacities, for CT score of 1. Image courtesy of American Journal of Roentgenology (AJR)
April 2, 2020 — Updated computed tomography (CT) scoring criteria that considers lobe involvement, as well as changes in CT findings, could quantitatively and accurately evaluate the progression of coronavirus disease (COVID-19) pneumonia, according to an open-access article in the American Journal of Roentgenology (AJR).
"The earlier that COVID-19 is diagnosed and treated, the shorter the time to disease resolution and the lower the highest and last CT scores are," concluded lead author Guoquan Huang of Wuhu Second People's Hospital in China.
Assigning CT scores to 25 patients according to CT findings and lung involvement, Huang and colleagues recorded the time from symptom onset to diagnosis and treatment for each patient. Patients with COVID-19 were divided into two groups: (patients for whom this interval was 3 days) and group 2 (those for whom the interval was > 3 days).
Using a Lorentzian line-shape curve to show the variation tendency during treatment, the fitted tendency curves for group 1 and group 2 were significantly different. Peak points showed that the estimated highest CT score was 10 and 16 for each group, respectively, and the time to disease resolution was 6 and 13 days, respectively.
The Mann-Whitney test showed that the last CT scores were lower for group 1 than for group 2 (p = 0.025), although the chi-square test found no difference in age and sex between the groups. The time from symptom onset to diagnosis and treatment had a positive correlation with the time to disease resolution (r = 0.93; p = 0.000), as well as with the highest CT score (r = 0.83; p = 0.006).
"Sequential chest CT examinations enable qualitative investigation of alterations in COVID-19 infection during the course of treatment," Huang explained.
Because previously proposed CT scoring criteria regarding lobe involvement gave no consideration to changes in CT features (i.e., the change from observation of GGO to a crazy-paving pattern and then consolidation), Huang et al. suggest that such a rubric is not sufficiently accurate to assess the progression of pneumonia.
"In the present study," wrote Huang, "we propose a new version of CT scoring criteria that considers both lobe involvement and changes in CT findings, in an attempt to more comprehensively evaluate COVID-19 pneumonia on sequential chest CT examinations."
For more information: www.arrs.org
Related Coronavirus Content:
Two Studies Use SIRD Model to Forecast COVID-19 Spread
Radiology Publishes First Case of COVID-19 Encephalopathy
VIDEO: Use of Telemedicine in Medical Imaging During COVID-19
VIDEO: How China Leveraged Health IT to Combat COVID-19
CDRH Issues Letter to Industry on COVID-19
Qure.ai Launches Solutions to Help Tackle COVID19
ASRT Deploys COVID-19 Resources for Educational Programs
Study Looks at CT Findings of COVID-19 Through Recovery
VIDEO: Imaging COVID-19 With Point-of-Care Ultrasound (POCUS)
The Cardiac Implications of Novel Coronavirus
CT Provides Best Diagnosis for Novel Coronavirus (COVID-19)
Radiology Lessons for Coronavirus From the SARS and MERS Epidemics
Deployment of Health IT in China’s Fight Against the COVID-19 Epidemic
Emerging Technologies Proving Value in Chinese Coronavirus Fight
Radiologists Describe Coronavirus CT Imaging Features
Coronavirus Update from the FDA
CT Imaging of the 2019 Novel Coronavirus (2019-nCoV) Pneumonia
CT Imaging Features of 2019 Novel Coronavirus (2019-nCoV)
Chest CT Findings of Patients Infected With Novel Coronavirus 2019-nCoV Pneumonia


 May 26, 2026
May 26, 2026 









