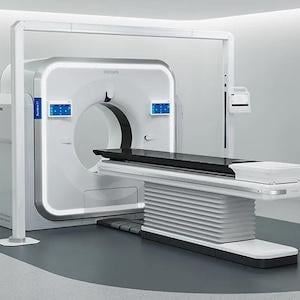
The Rembra platform of systems also extends the role of CT across both diagnostic radiology and radiation therapy, helping to enable more connected workflows and a coordinated approach to patient care. (Photo: Royal Philips)
April 23, 2026 — Royal Philips has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Rembra platform of scanning systems, including Rembra CT, Rembra RT and Areta RT.
The newly cleared systems are designed to help healthcare providers respond to growing demand for imaging and radiation therapy planning by helping to improve speed, efficiency, and access to high-quality imaging. As imaging volumes and clinical complexity continue to rise, healthcare providers need solutions that can help them work more efficiently while maintaining confidence in clinical decisions.
The Rembra platform of systems also extends the role of CT across both diagnostic radiology and radiation therapy, helping to enable more connected workflows and a coordinated approach to patient care—from rapid assessment in acute settings to precise cancer treatment planning.
“As healthcare systems manage increasing demand and complexity, imaging plays a critical role in enabling timely and informed clinical decisions,” said Dan Xu, Business Leader of CT at Philips. “With the Rembra platform, we are redefining what clinicians can expect from CT, combining speed, scalability, and precision to expand access to high-quality imaging while supporting confident diagnosis and highly accurate treatment planning.”
With the largest-in-class 85 cm bore, Rembra CT is purpose-built for the front lines of care, enabling ultra-fast, high-throughput imaging in demanding clinical environments, supporting up to 270 exams per day1 in various settings including emergency departments, critical care, and interventional settings. Its advanced acquisition and reconstruction capabilities support rapid, high-quality imaging at scale, helping clinicians make timely decisions in critical situations without compromising diagnostic confidence.
Rembra RT and Areta RT bring these performance advantages into radiation therapy, where precision is critical. These systems support high-fidelity imaging for treatment planning with a wide 85 cm extended field of view2 and next generation 4DCT imaging capabilities, supporting more accurate targeting of tumors while helping protect healthy tissue. By streamlining simulation and planning workflows, they help reduce variability and improve efficiency, supporting more precise, personalized cancer care.
Together, the Rembra platform delivers a unified, high-performance CT ecosystem that spans the full care continuum—from urgent diagnosis to precision-guided therapy. It empowers healthcare providers to manage increasing demand with greater speed, consistency, and confidence while enabling more connected and efficient care delivery.
The FDA clearances further strengthen Philips’ comprehensive CT portfolio and reflect its continued focus on integrating advanced imaging technologies with intelligent and connected workflows. They also reflect Philips’ ongoing commitment to evolving its CT offering to address the changing needs of today’s imaging environments. By bringing together diagnostic and therapy solutions, Philips aims to support more efficient, coordinated care and improve the experience for both patients and care teams.
For more information about Rembra, please visit Philips - Rembra 728383 CT scanner.
Additional information on Philips
Sources
-
Based on a sixteen-hour day, with throughput testing with 203 patient scans/ 12 hours for Rembra with typical radiological profiles and protocols.
-
The Extended Field of View (EFOV) of 85 cm is intended solely for use in treatment preparation and the planning/simulation of radiation therapy. It cannot be used for diagnostic purposes. The water equivalent material external contour deviation of body system phantom positioned (partially) outside scan FOV with phantom edge adjacent to bore cover is within 1 mm in terms of mean Hausdorff distance compared to the true external contour.


 May 29, 2026
May 29, 2026 









