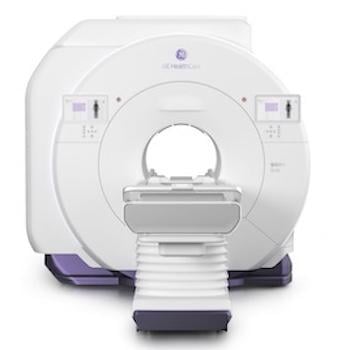
SIGNA Bolt. Photo: GE HealthCare
May 11, 2026 – At the International Society for Magnetic Resonance in Medicine (ISMRM) 2026 Annual Meeting, GE HealthCare is announcing advancements in research tools and MRI systems that aim to accelerate discovery, strengthen collaboration across research programs and turn innovation into meaningful clinical achievements.
GE HealthCare continues to invest in advanced MR technology with its next-generation SIGNA MR systems and research tools designed to help academic and clinical research programs move faster, work more collaboratively and push the boundaries of discovery. By combining high-performance hardware with advanced software and deep learning–based techniques, these innovations are intended to reduce scanning and examination times, improve image quality, and enable more consistent data across sites and studies.
The company's focus "is to enable the research ecosystem that helps researchers explore new questions, scale collaboration and translate discoveries more efficiently to broaden clinical impact,” said Kelly Londy, president and CEO, MR and Women’s Health at GE HealthCare. “From high-performance systems to collaboration resources for productive MR research, we are committed to giving researchers the tools they need to work together more effectively, explore new frontiers, and accelerate the path from discovery to patient experience.”
MRI AI-driven technologies
Expanding on GE HealthCare’s leadership in AI-enabled medical device 510(k) clearances,1 GE HealthCare announced the U.S. FDA 510(k) pending status of its latest deep-learning acceleration technique, Sonic DL for 2D imaging beyond cardiac cine2. Built on the foundation of previous Sonic DL technology for cardiac cine and 3D imaging, this latest solution will mean up to 85% of MR exams will benefit from the deep-learning acceleration of Sonic DL, which is further enhanced by compatibility with AIR Recon DL for 2D and 3D imaging and will further enhance image quality and Signal-to-Noise Ratio (SNR) of accelerated exams.
At the foundation of GE HealthCare’s latest MR capabilities is SIGNA One3, a new AI-powered, next-gen workflow ecosystem designed to simplify complexity, improve efficiency and enable more consistent imaging across clinical and research environments. By addressing inefficiencies throughout the MRI workflow, SIGNA One helps increase throughput, reduce variability, and improve the experience for researchers, technologists and patients alike.
SIGNA One integrates a suite of core workflow solutions, including an intuitive and AI-guided user experience designed to minimize training time and support users of varying experience levels, as well as one-click switching between clinical and research modes4 within an exam for seamless integration of research protocols within clinical workflows. Together, these capabilities are designed to support more seamless collaboration and reproducibility — critical enablers for advanced MR research programs.
Built on the SIGNA One platform, SIGNA Bolt5, GE HealthCare’s next-generation 510(k) cleared 3T MRI system, extends these capabilities with powerful performance grounded in deep learning and a focus on sustainability. The system is designed to streamline end-to-end imaging pathways, support precision diagnostics, and maintain low energy consumption and operational costs — reinforcing GE HealthCare’s commitment to advancing MR research and clinical innovation responsibly.
The new SIGNA Sprint with Freelium6 offers helium-free technology, eliminating vent pipe requirements, without compromising clinical or
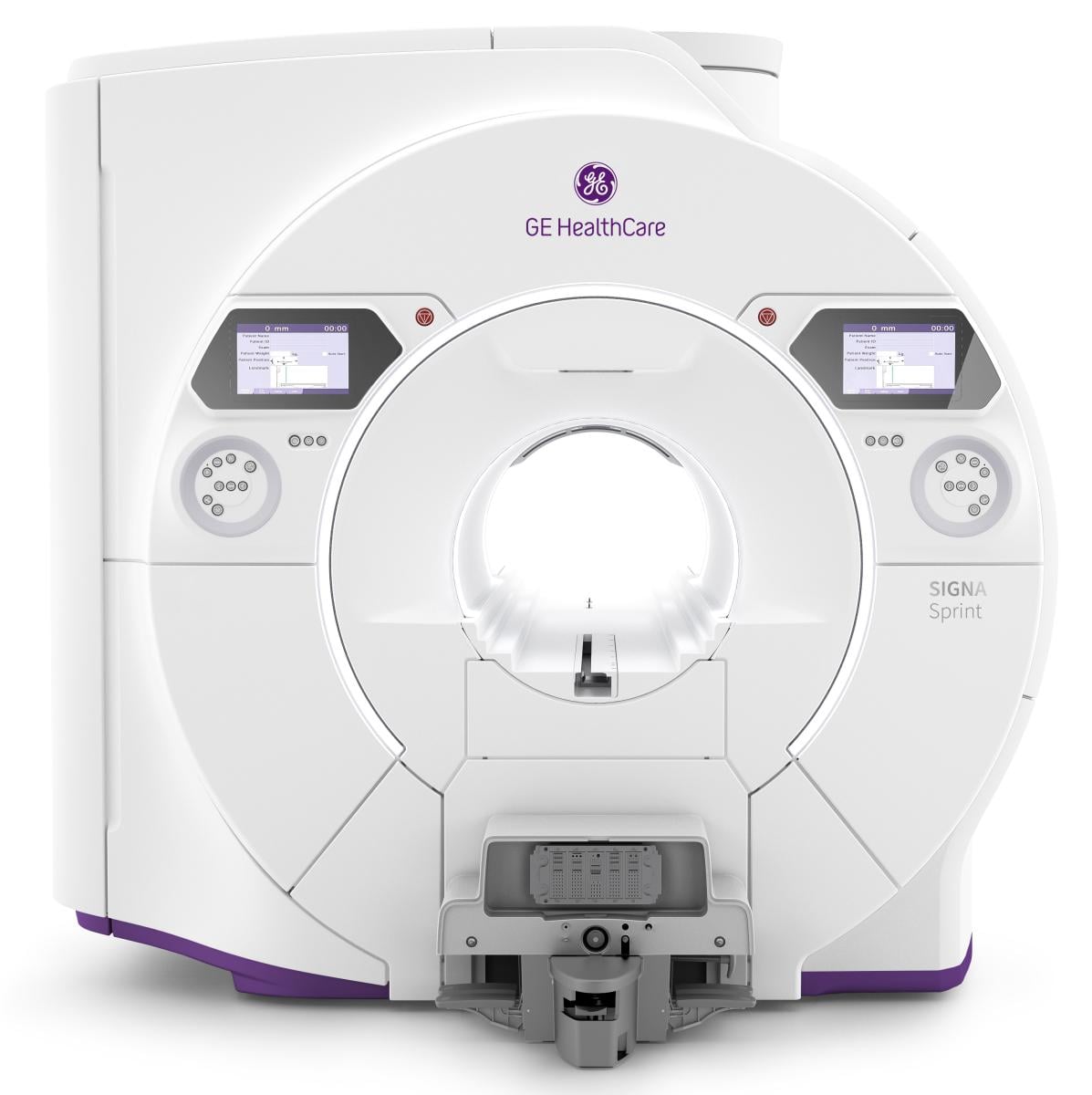
operational efficiency. With less than 1% helium usage7, SIGNA Sprint with Freelium provides helium independence without driving higher power consumption requirements or sacrificing image quality.
Additionally, AIR Recon DL, GE HealthCare’s deep-learning reconstruction technology, is expanding with the addition of support for Zero Echo Time (ZTE) and Silenz low-acoustic noise imaging8. The basis for the MR bone imaging technique, oZTEo, ZTE imaging can provide images similar to CT — but without ionizing radiation — as well as novel applications such as lung imaging. AIR Recon DL will enable reduced scan time and improve the image quality of ZTE exams, while also enabling the potential for improved 3D volume rendering.
Research Collaboration
GE HealthCare also will introduce SIGNA Studio9, a suite of software development and collaboration resources for productive, impactful and efficient MR research. SIGNA Studio components are designed to make research tools more accessible and faster for researchers to deploy.
Building on early research collaborations, GE HealthCare announces two additional installations of the investigational MAGNUS prototype10, a head-only MR scanner designed to explore advancements in neuroscience, at both King’s College London (KCL) and West China Hospital to push the boundaries of microstructural imaging, advanced diffusion and functional techniques, amplified by GE HealthCare’s latest deep-learning technology including Sonic DL and AIR Recon DL.
“This installation at King’s College London represents an important step forward for neuroscience research,” said Dr. Steve Williams, Professor of Imaging Sciences at King’s College London, who will present during GE HealthCare’s lunch symposium at ISMRM. “This platform gives us the ability to investigate aspects of brain structure and function that were previously out of reach, helping advance our understanding of neurological disease and opening new research possibilities.”
GE HealthCare also continues to invest in advancing the neuro care pathway, spanning research, imaging, and clinical decision support. This includes the integration of icometrix and its icobrain platform to strengthen quantitative brain MRI analysis and support more personalized neurological care, particularly in areas such as Alzheimer’s disease.
References
-
U.S. Food and Drug Administration. “Artificial Intelligence-Enabled Medical Devices.” https://www.fda.gov/medical-devices/software-medical-device-samd/artifi….
-
Sonic DL for 2D imaging is 510(k) pending with the U.S. FDA. Not CE marked. Not available for sale in all regions.
-
SIGNA One Interface features are offered on select GE HealthCare MRI Systems. The features are 510(k) cleared. Not CE Marked. Not available in all markets.
-
Research mode is available as an option and not intended for clinical use. Research operation may require observation of national regulations.
-
SIGNA Bolt is 510(k) cleared. Not CE Marked. Not available in all markets.
-
SIGNA Sprint with Freelium is a sealed configuration of SIGNA Sprint Select. SIGNA Sprint Select is 510(k) cleared. Not CE Marked. Not available in all markets.
-
Compared to conventional magnets.
-
AIR Recon DL for ZTE imaging is 510(k) pending with the U.S. FDA. Not CE marked. Not available for sale in all regions.
-
SIGNA Studio is a set of tools for MR research and development use only. Not intended for clinical use. Research use may be subject to additional requirements in each country or jurisdiction. Not a product. Not for sale in any region.
-
Technology in development that represents ongoing research and development efforts. Not 510(k) cleared and not CE marked. Not for sale. Intended for investigational use only.


 May 12, 2026
May 12, 2026 









