October 7, 2016 — Toshiba America Medical Systems Inc. announced it will be displaying a wide array of diagnostic imaging technologies at the 2016 Radiological Society of North America (RSNA) annual meeting, Nov. 27-Dec. 2 in Chicago.
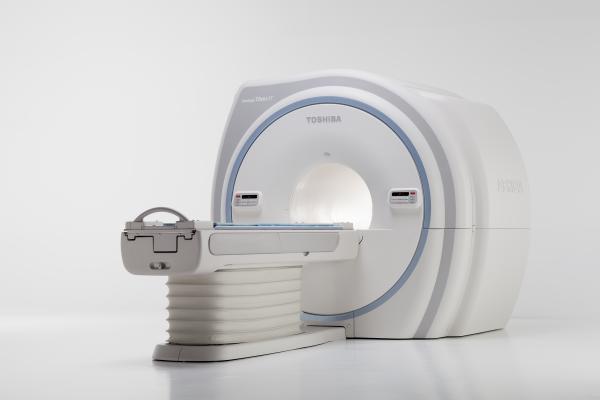
Helping healthcare providers diversify their magnetic resonance imaging (MRI) offerings and reduce their exam times, Toshiba will showcase its newest MR system at RSNA — the Vantage Titan/cS Edition, pending U.S. Food and Drug Administration (FDA) 510(k) clearance. The 1.5T system offers a suite of innovative applications to help improve patient comfort and workflow. To ensure a positive patient experience, the system features a 71 cm bore, feet-first exams and Toshiba’s Pianissimo noise-reduction technology. Helping clinicians increase workflow, Toshiba’s Vitrea Extend application permits clinicians to complete post-processing at a separate workstation, potentially allowing more patients to be scanned in the MR system. The system also offers an optional CardioLine+ application and SureVOI Cardiac technology to automatically detect anatomical landmarks for fast and accurate cardiac MR exams.
Toshiba is also introducing enhancements to its Vantage Titan 3T MR system to help clinicians improve the reproducibility and efficiency of neurological and musculoskeletal (MSK) imaging. Toshiba’s Saturn Gradient and Atlas Speeder coils have been upgraded to enable high-quality images of soft tissue and cartilage to help clinicians make diagnoses and provide rapid, effective treatment. These technology improvements help in neurological exams, which is particularly important with concussion and brain trauma cases. The system also accommodates a large range of clinical needs by pairing high-quality images with a variety of patient-friendly features, including a 71-cm patient aperture and Toshiba’s Pianissimo noise-reduction technology, among many others.
For more information: www.medical.toshiba.com


 May 19, 2026
May 19, 2026 









