December 27, 2012 — A new way to use MRI scans may help determine whether dementia is Alzheimer’s disease or another type of dementia, according to new research published in the Dec. 26, 2012, online issue of Neurology, the medical journal of the American Academy of Neurology.
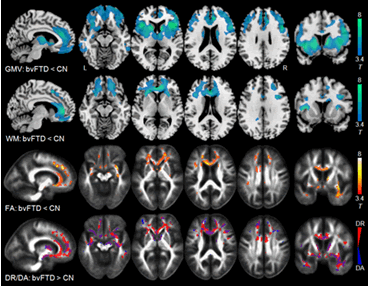
Alzheimer’s disease and frontotemporal lobar degeneration (FTLD) often have similar symptoms, even though the underlying disease process is much different.
“Diagnosis can be challenging,” said study author Corey McMillan, Ph.D., of the Perelman School of Medicine and Frontotemporal Degeneration Center at the University of Pennsylvania and a member of the American Academy of Neurology. “If the clinical symptoms and routine brain MR are equal, an expensive positron emission tomography (PET) scan might be needed. Or, a lumbar puncture, which involves inserting a needle into the spine, would be needed to help make the diagnosis. Analysis of the cerebrospinal fluid gives us reliable diagnostic information, but this is not something patients look forward to and is also expensive. Using this new MRI method is less expensive and definitely less invasive.”
The study involved 185 people who had been diagnosed with a neurodegenerative disease consistent with Alzheimer’s disease or FTLD and had a lumbar puncture and a high resolution MRI. Of the 185, the diagnosis was confirmed in 32 people either by autopsy or by determining that they had a genetic mutation associated with one of the diseases.
Researchers used the MRIs to predict the ratio of two biomarkers for the diseases in the cerebrospinal fluid, the proteins tau and beta-amyloid. The MRI prediction method was 75 percent accurate at identifying the correct diagnosis in those with pathology-confirmed diagnoses and those with biomarker levels obtained by lumbar punctures, which shows similar accuracy of the MRI and lumbar puncture methods.
“Developing a new method for diagnosis is important because potential treatments target the underlying abnormal proteins, so we need to know which disease to treat,” McMillan said. “This could be used as a screening method and any borderline cases could follow up with the lumbar puncture or PET scan. This method would also be helpful in clinical trials where it may be important to monitor these biomarkers repeatedly over time to determine whether a treatment was working, and it would be much less invasive than repeated lumbar punctures.”
The study was supported by the Wyncote Foundation and the National Institutes of Health.
For more information: www.aan.com


 May 27, 2026
May 27, 2026 









