January 7, 2022 — A Case Western Reserve University-led team of scientists has used Artificial Intelligence (AI) to ...
January 6, 2022 — Royal Philips announced new additions to its CT imaging portfolio at the Radiological Society of North ...
January 6, 2022 — Staffordshire University health experts claim there is an urgent need for global telehealth guidelines ...
AT A GLANCE Organization: Expert Radiology Management Services, LLC Specialty: Subspecialty teleradiology — neuro and ...
Jef Williams, MBA, PMP, CIIP, managing partner, Paragon Consulting Partners LLC, explains trends he saw at the 2021 ...
January 5, 2022 — According to an article in ARRS’ American Journal of Roentgenology (AJR), brain volume measurements in ...
January 5, 2022 — A new neuroimaging technique can detect biochemical changes in the brains of people with multiple ...
Radiology departments have many different needs and face a wide variety of challenges that can impact their departments ...
January 5, 2022 — The Radiological Society of North America’s 107th Scientific Assembly and Annual Meeting (RSNA 2021) ...
January 5, 2022 — Hamburg, Germany, based Mindpeak and Spokane, Wash., based Gestalt Diagnostics have achieved a major ...
January 5, 2022 — Abbott announced that the U.S. Food and Drug Administration (FDA) has approved new expanded magnetic ...
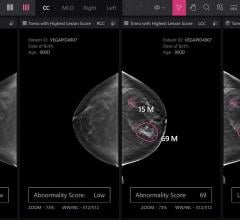
Despite decades of progress in breast imaging, one challenge continues to test even the most skilled radiologists ...
January 4, 2022 — The future of ultrasound is happening — fast — and it doesn’t cost that much more than an iPhone. At ...
January 4, 2022 — Rome Radiology Group (RRG), based in Rome, Ga., became the first member of the national Strategic ...
January 3, 2022 — IBA (Ion Beam Applications S.A., EURONEXT), the world leader in particle accelerator technology and ...
Bayer Radiology’s Barbara Ruhland and Thom Kinst discuss how radiology departments can address the many different ...
January 3, 2022 — The "Computed Tomography (CT) Scanners Devices and Equipment Global Market Report 2021: COVID-19 ...
Wishing you a healthy and happy New Year from the ITN team. Look for many exciting new changes to come in 2022!
December 31, 2021 — International medical imaging IT and cybersecurity company Sectra will provide its enterprise ...
December 31, 2021 — RAYUS Radiology has opened a new outpatient-based advanced diagnostic imaging center in Auburn ...
December 30, 2021 — IBA (Ion Beam Applications S.A), a world leader in particle accelerator technology, announced the ...
December 30, 2021 — The "Global Contrast Media Market Size, Share & Trends Analysis Report by Product Type (Iodinated ...
December 29, 2021 — Researchers at the University of Surrey have identified key design rules for making curved X-ray ...
December 29, 2021 — The global nuclear imaging devices and equipment market is expected to grow from $2.45 billion in ...


 January 07, 2022
January 07, 2022 
![Royal Philips announced new additions to its CT imaging portfolio at the Radiological Society of North America (RSNA) annual meeting. The new CT 5100 – Incisive – features CT Smart Workflow [1], a comprehensive suite of artificial intelligence* (AI) enabled capabilities designed to accelerate CT workflows, enhance diagnostic confidence, and maximize equipment up-time, helping imaging services to enhance patient outcomes, improve department efficiency, reduce operational costs, and meet ambitious financial o](/sites/default/files/styles/content_feed_medium/public/Screen%20Shot%202022-01-06%20at%209.55.32%20AM.png?itok=HEydSu0e)










![IBA (Ion Beam Applications S.A., EURONEXT), the world leader in particle accelerator technology and the world’s leading provider of proton therapy solutions for the treatment of cancer, announced a new contract for the installation of a Proteus ONE[1] proton therapy solution with Proton International Arkansas LLC – a joint venture formed by Proton International, the University of Arkansas for Medical Sciences (UAMS), Baptist Health, and Arkansas Children’s.](/sites/default/files/styles/content_feed_medium/public/IBA_Arkansas.jpeg?itok=oKTTWRxs)