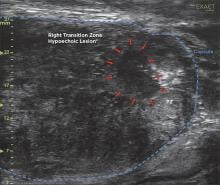
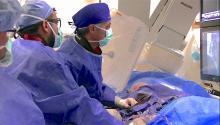
Historically when a patient had an elevated PSA (prostate specific antigen) test their urologist would take the next step — a prostate biopsy to determine if the increased PSA level was due to cancer or another condition such as infection or benign prostatic hyperplasia. These biopsies were performed using standard ultrasound to identify the prostate.
© Copyright Wainscot Media. All Rights Reserved.
Subscribe Now