
MRI Targeted biopsy is performed using cognitive fusion more easily with anatomical guidance based on the radiology report. MRI targets can be identified quickly in real-time along with micro-ultrasound targets, which may have been missed on MRI. Image courtesy of Exact Imaging
Historically when a patient had an elevated PSA (prostate specific antigen) test their urologist would take the next step — a prostate biopsy to determine if the increased PSA level was due to cancer or another condition such as infection or benign prostatic hyperplasia. These biopsies were performed using standard ultrasound to identify the prostate. Unfortunately, standard ultrasound is not able to provide much detail of the tissue within the prostate.
Doing a standard ultrasound to detect prostate cancer was like playing darts blindfolded. It required the doctor to take an average of 12 random samples in the hopes of finding the cancer. This practice often misses the cancer or stops short of the edge of the abnormal tissue leading to the patient receiving a benign or negative test result when they actually have prostate cancer, or a diagnosis of low-risk cancer when high-risk aggressive disease is actually present.
Random or systematic biopsy has led to a large number of men undergoing this invasive and painful procedure multiple times over several years before their cancer is properly diagnosed and treated. Aside from the anxiety and suffering it causes, this delay is unfortunate because prostate cancer is nearly 100 percent curable when caught early.
Multi-parametric Magnetic Resonance Imaging
Over the past decade, multi-parametric magnetic resonance imaging (mpMRI) has emerged, which has helped men avoid unnecessary biopsies as well as improve biopsy accuracy. For this reason, the National Comprehensive Cancer Network (NCCN) now recommends that physicians consider mpMRI prior to a prostate biopsy (NCCN Clinical Practice Guidelines in Oncology V1.2019, 2019).1
However, mpMRI has several drawbacks, including inadequate training and quality assurance for radiologists who do not specialize in prostate cancer, unreliable insurance reimbursement and the use of gadolinium-based contrast agents. Further, for some patients who have decreased kidney function, are claustrophobic or have a pacemaker, MRI may not be possible.
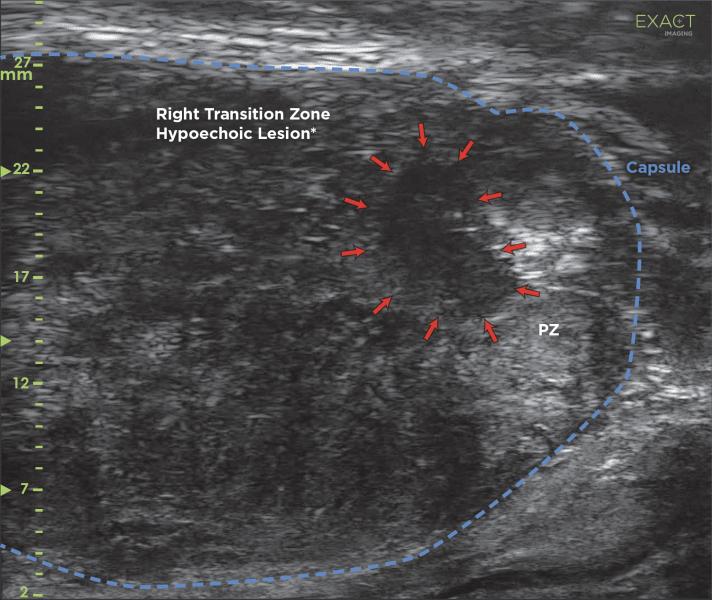
Micro-ultrasound is a new, improved ultrasound technology that operates at 29 MHz compared to 7.5 MHz in standard ultrasound, which leads to a 300 percent improvement in resolution. Micro-ultrasound offers a dramatically higher resolution down to 70 microns, which is the thickness of a human hair, and the size of a typical prostatic duct. This higher resolution allows micro-ultrasound to detect prostate cancer with very high sensitivity, which was not possible using conventional ultrasound (Abouassaly, et al, 2019; Pavlovich et al., 2013).2,3
What is particularly exciting about this technology is that it can be used just like regular ultrasound, in real-time by the physician performing the biopsy directly in the clinic. This speeds up screening, simplifies care, is less expensive and does not require any contrast injections.
Micro-ultrasound Technology
With micro-ultrasound technology, doctors have access to live images that better visualize suspicious areas for improved detection of the cancer and estimate of how aggressive the cancer will be. The urologist’s ability to view the patient’s anatomy, detect suspicious areas, differentiate between benign and cancerous tissue and target specific areas for biopsy has caused micro-ultrasound to be adopted very quickly by experts within the prostate cancer field.
Recent new work has shown micro-ultrasound to be even better than the more expensive mpMRI for detecting prostate cancer. A study comparing micro-ultrasound to mpMRI, led by Laurence Klotz, M.D., chief of urology at Sunnybrook Health Sciences Center and professor of surgery at the University of Toronto, was published in the Canadian Urology Association Journal (Klotz et al., 2020) analogous to multiparametric magnetic resonance imaging.4 The study reports on 1,040 men who received both mpMRI and micro-ultrasound imaging prior to biopsy. This large-scale, real-world study involved 11 institutions in the United States, Canada and Europe, including world leaders in mpMRI. The goal was to compare the sensitivity and negative predictive value (NPV) of each imaging test to predict whether cancer would be found in a biopsy.
These are important metrics because they can help patients make an informed decision about whether or not to have a biopsy. In particular, NPV is the rate of finding only benign tissue in a biopsy following a negative or non-suspicious imaging result. The higher the NPV, the higher the patient’s confidence in avoiding a biopsy when the imaging is negative. In this study, micro-ultrasound demonstrated a higher NPV than mpMRI (85 percent vs. 77 percent, p=0.04). Micro-ultrasound also demonstrated a higher sensitivity (94 percent vs. 90 percent, p=0.03), which is the percentage of all cancers that were detected.
More importantly, these benefits were realized without any decrease in specificity or positive predictive value, suggesting both techniques would allow roughly the same number of men to avoid biopsy. For the average man in this study, a negative micro-ultrasound reduced the risk of finding cancer by 62 percent, while a negative MRI reduced the same risk by 41 percent.
Positive Outlook for Patients
What does this mean for patients? It may mean that instead of the urologist referring a patient out for mpMRI, he can perform a micro-ultrasound scan in the office, which takes approximately 15 minutes. Together with other risk factors and biomarkers, the results will help patients feel more confident in their decision to either proceed with a biopsy or to postpone it.
As with any clinical study, further large-scale investigation is warranted. However, the authors conclude that micro-ultrasound is an appealing alternative to mpMRI for detecting prostate cancer.
Brian Wodlinger, Ph.D., is vice president, engineering and clinical at Exact Imaging. Prior to Exact Imaging, Wodlinger held research positions at the University of Pittsburgh Medical Center and Case Western Reserve University. This research won Popular Mechanics Top Breakthroughs of the Year award and has been featured in the popular media on platforms such as CBS’s “60 Minutes.” He holds eight patents in the medical devices space, has designed and executed more than 13 clinical trials and has authored more than 18 peer-reviewed research articles in prestigious journals such as The Lancet and PLoS ONE.
References:
1. NCCN Clinical Practice Guidelines in Oncology V1.2019. (2019). Prostate Cancer Early Detection Recommendations. National Comprehensive Cancer Network, Inc. https://www.nccn.org/professionals/physician_gls/pdf/prostate_detection.pdf
2. Abouassaly, R., Klein, E. A., El-Shefai, A., & Stephenson, A. (2019). Impact of using 29 MHz high-resolution micro-ultrasound in real-time targeting of transrectal prostate biopsies: initial experience. World Journal of Urology, 0123456789, 1–6. https://doi.org/10.1007/s00345-019-02863-y
3. Pavlovich, C. P., Cornish, T. C., Mullins, J. K., Fradin, J., Mettee, L. Z., Connor, J. T., Reese, A. C., Askin, F. B., Luck, R., Epstein, J. I., & Burke, H. B. (2013). High-resolution transrectal ultrasound: Pilot study of a novel technique for imaging clinically localized prostate cancer. Urologic Oncology. https://doi.org/10.1016/j.urolonc.2013.01.006
4. Klotz, L., Lughezzani, G., Maffei, D., Sanchez, A., Pereira, J. G., Staerman, F., Cash, H., Luger, F., Lopez, L., Sanchez-Salas, R., Abouassally, R., Shore, N. D., & Eure, G. (2020). Comparison of micro-ultrasound and multiparametric magnetic resonance imaging for prostate cancer: A multicenter, prospective analysis. Canadian Urological Association Journal, 15(1), 1–13. https://doi.org/10.5489/cuaj.6712



 May 20, 2026
May 20, 2026 









