June 22, 2021 — NorthStar Medical Radioisotopes, LLC, a global innovator in the development, production and commercialization of radiopharmaceuticals used for therapeutic applications and medical imaging, and Clarity Pharmaceuticals, a clinical stage radiopharmaceutical company focused on the treatment of serious disease, announced the signing of a Master Supply Agreement for the therapeutic radioisotope copper-67 (Cu-67). Under the agreement, NorthStar will supply Cu-67 exclusively to Clarity Pharmaceuticals as an active pharmaceutical ingredient used to support Clarity’s Targeted Copper Theranostics (TCT) programs.
Cu-67 is a beta-emitting radioisotope with clinical applications as a radiopharmaceutical to directly target and deliver therapeutic doses of radiation to destroy cancer cells in patients with serious disease.
“Previously, the lack of an effective copper chelating technology has limited the clinical development of Cu-67 products and subsequent commercial production of Cu-67,” said Stephen Merrick, President and Chief Executive Officer of NorthStar Medical Radioisotopes. “Clarity Pharmaceuticals’ unique copper-chelating technology has enabled it to advance its product pipeline into a range of theranostic clinical trials that use copper-64 (Cu-64) for diagnostic imaging and Cu-67 as therapy. NorthStar is addressing the need for reliable Cu-67 radioisotope supply by advancing towards commercial-scale production. Our efforts use the proven expertise and innovative approach demonstrated in the successful development and commercialization of U.S.-produced, non-uranium molybdenum-99 (Mo-99) to generate technetium-99m. NorthStar is pleased to sign this agreement with Clarity Pharmaceuticals, and we look forward to supporting its plans for further clinical development and commercialization of Cu-67 based therapeutic radiopharmaceuticals to improve the lives of patients with serious disease.”
“Consistent with other NorthStar production processes, the Cu-67 production process is also environmentally friendly and non-uranium based,” said James T. Harvey, PhD, Senior Vice President and Chief Science Officer of NorthStar Medical Radioisotopes. “Cu-67 has an optimal half-life for a therapeutic radiopharmaceutical, is produced domestically in the United States without a nuclear reactor, has no long-lived contaminants or bi-products from the manufacturing process, and its starting material, zinc, is readily available. As opposed to other therapeutic isotopes in the market and under development, Cu-67 is ideally suited for commercial production to meet the growing demand for radiopharmaceuticals in large disease indications and we look forward to meeting Clarity’s anticipated future demands as it develops and commercializes its suite of products.”
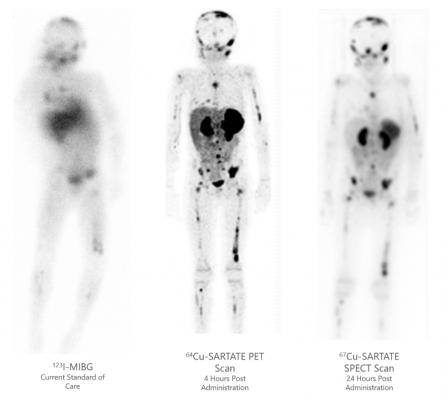
“Clarity leads the world in the development and commercialization of TCT,” said Alan Taylor, PhD, Executive Chairman of Clarity Pharmaceuticals. “Access to large, commercial supply of Cu-67 at a suitable price-point enables us to apply our TCT approach not only to rare diseases such as neuroblastoma in children, but also other cancers with very large patient populations such as prostate and breast cancers. In essence, the availability of large scale production of Cu-67 in the United States will increase the speed of Clarity’s clinical trials across our SARTATE, SAR-bisPSMA, SAR-Bombesin and discovery programs and will help us to achieve our ultimate goal of better treating children and adults with cancer.”
About Copper-67 (Cu-67)
Copper-67 (Cu-67) is a short-range, beta-emitting radioisotope that is attractive for medical purposes due to its ability to carry sufficient radiation energy to cause cell death in targeted cells while having a sufficiently short half-life to limit unwanted radioactivity in patients. Cu-67 is being investigated for therapeutic purposes across a wide range of adult and childhood cancers. Potential radiotherapeutic targets include prostate cancer, breast cancer, neuroendocrine tumors (NETs), neuroblastoma, glioma, lymphoma, ovarian, and bladder cancers. A chelator, which strongly binds Cu-67 to the targeting agent, is required to develop safe and effective targeted therapies. Clarity has successfully developed a highly specific and highly stable chelator for copper isotopes and is progressing a range of radiopharmaceuticals based on its proprietary SAR Technology Platform. NorthStar is developing a proprietary process for commercial-scale production of Cu-67 to meet demand for clinical research and treatment purposes.
For more information: www.northstarnm.com
Related Content:
How Nuclear Fusion is Revolutionizing Medical Isotope Production
FDA Approves Additional Molybdenum-99 (Mo-99) Filling Lines at NorthStar Medical Radioisotopes
Medical Isotope Industry Opposes Export of Highly Enriched Uranium for IRE
IBA and NorthStar Medical Radioisotopes Expand Collaboration to Enable Global Availability of Tc-99m


 May 06, 2026
May 06, 2026 









