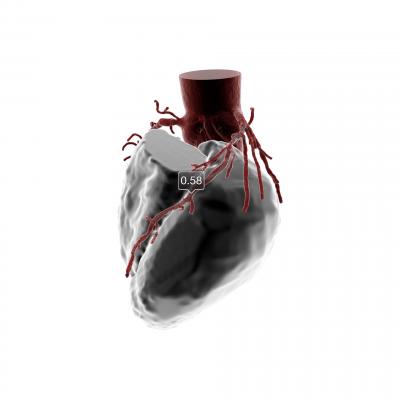
February 1, 2018 – HeartFlow announced that seven new commercial payers issued positive medical policies covering the use of the HeartFlow FFRct (fractional flow reserve computed tomography) Analysis following a coronary computed tomography angiogram (CCTA). The HeartFlow Analysis is a non-invasive technology that creates a personalized 3-D model of the heart and simulates blood flow to help clinicians diagnose and treat patients with suspected heart disease.
The positive medical policies were issued by Cigna; Florida Blue; the Blue Cross and Blue Shield plans of Michigan, North Carolina, and Vermont; Highmark Inc.; and Independence Blue Cross. Commercial coverage for the HeartFlow Analysis is now available for approximately 130 million patients, in addition to the Centers for Medicare and Medicaid Services (CMS) recent decision to assign a New Technology APC payment for the HeartFlow FFRct Analysis for more than 55 million Medicare beneficiaries.
Coronary artery disease (CAD) affects 16.8 million Americans and develops when the coronary arteries narrow, reducing blood flow to the heart and causing chest pain (angina), heart attack (myocardial infarction) and death. The HeartFlow Analysis is the first and only commercially available non-invasive technology, according to the company, to provide insight into both the extent of CAD and the impact of the disease on blood flow to the heart.
For more information: www.heartflow.com


 May 19, 2026
May 19, 2026 









