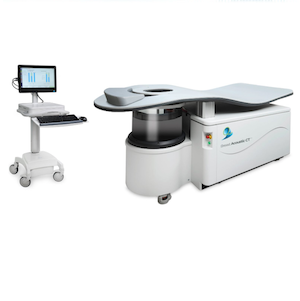
Photo: QT Imaging
March 10, 2026 — QT Imaging Holdings has received U.S. Food and Drug Administration (FDA) 510(k) clearance for an updated configuration of its Breast Acoustic CT scanner, the company’s 3D ultrasound tomographic breast imaging system. The newly cleared enhanced configuration improves visualization and expands coverage of posterior breast tissue.
“The updated configuration cleared by the FDA incorporates a tilted transmitter geometry that improves tomographic imaging coverage of breast tissue located near the chest wall, which is an area that can be extremely challenging to capture,” said Bilal Malik, PhD, Chief Science Officer of QT Imaging. “This design delivers more complete coverage and improved overall diagnostic utility by optimizing the scanner’s ability to include posterior breast tissue that is often difficult to image with standard approaches. We remain dedicated to developing and refining innovative technologies that provide greater precision and confidence to deliver the best outcomes for women regardless of breast density.”
The QTI Breast Acoustic CT scanner generates both reflection-mode and transmission-mode ultrasound data to reconstruct 3D tomographic images. Its proprietary software quantifies fibroglandular tissue volume (FGV) and the ratio of fibroglandular tissue to total breast volume (TBV), providing valuable insight for breast health assessment and monitoring.
To learn more about the Breast Acoustic CT scanner go to www.qtimaging.com/breast-acoustic-ct-scanner.


 May 19, 2026
May 19, 2026 









