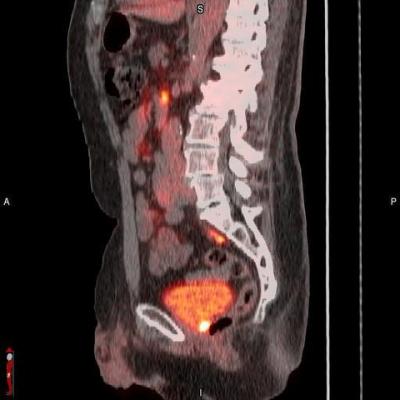
POSLUMA (flotufolastat F 18) PET/CT image showing uptake in the prostate bed, consistent with recurrent prostate cancer Photo courtesy of Blue Earth Diagnostics
September 27, 2023 — Blue Earth Diagnostics, a Bracco company and recognized leader in the development and commercialization of innovative PET radiopharmaceuticals, announced presentations on POSLUMA (flotufolastat F 18) injection (formerly known as 18F-rhPSMA-7.3) at the upcoming American Society for Therapeutic Radiology and Oncology (ASTRO) 2023 Annual Meeting, to be held in San Diego, Calif., from October 1 to 4, 2023. POSLUMA is indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer with suspected metastasis who are candidates for initial definitive therapy or with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level.
“PET imaging with POSLUMA reveals clinical information crucial to decision-making for men with prostate cancer, and we are excited to share further information with the radiation oncology community at ASTRO 2023,” said David E. Gauden, D.Phil., Chief Executive Officer of Blue Earth Diagnostics. “Phase 3 results from the SPOTLIGHT trial demonstrated the ability of POSLUMA to detect recurrent disease even at low prostate specific antigen (PSA) levels, and Dr. Ashesh Jani’s oral presentation will detail further information about its diagnostic performance at PSA levels <1 ng/mL. Dr. Phillip Kuo will present additional results from the Phase 3 LIGHTHOUSE trial that evaluated POSLUMA performance in newly diagnosed patients having high/very high risk prostate cancer and negative results with conventional imaging. Blue Earth Diagnostics will also host an Industry-Expert Theater event, ‘POSLUMA: Precision PET Imaging that is Truly Revealing’.”
Details of selected oral and poster presentations by Blue Earth Diagnostics and its collaborators are listed below.
HIGHLIGHTED SCIENTIFIC PRESENTATIONS
Monday, October 2, 2023
|
Oral presentation |
||
|
Title: |
|
Detection Rate of 18F-rhPSMA-7.3 PET in Patients with Suspected Prostate Cancer Recurrence at PSA Levels <1 ng/mL: Data from the Phase 3 SPOTLIGHT Study |
|
Presenter: |
|
Ashesh B. Jani, MD, FASTRO, Department of Radiation Oncology, Winship Cancer Institute of Emory University, Atlanta, Ga., for the SPOTLIGHT Study Group |
|
Session Type: |
|
Oral |
|
Session Title: |
|
SS11 - GU 3 Novel Prognostication Techniques for Prostate Cancer |
|
Presentation Time: |
|
3:30 – 3:37 PM PT |
|
Location: |
|
Room 6 D/E |
|
Presentation No.: |
|
160 |
Tuesday, October 3, 2023
|
Poster presentation |
||
|
Title: |
|
Diagnostic Performance of 18F-rhPSMA-7.3 PET in Men with Newly Diagnosed High-risk Prostate Cancer and Negative Conventional Imaging |
|
Presenter: |
|
Phillip H. Kuo, MD, Ph.D., Departments of Medical Imaging, Medicine, and Biomedical Engineering, University of Arizona, Tucson, Ariz., on behalf of Gary A. Ulaner, MD, Ph.D., Hoag Family Cancer Institute, Irvine, Calif. and University of Southern California, Los Angeles, Calif., for the LIGHTHOUSE Study Group |
|
Session Title: |
|
PQ 06 – Poster Q&A 06 – Session 06 – Genitourinary Cancer, Patient Safety and Nursing |
|
Presentation Time: |
|
2:30 – 3:45 PM PT |
|
Location: |
|
Hall B1 |
|
Presentation No.: |
|
2972 |
Blue Earth Diagnostics invites participants at the 2023 ASTRO Annual Meeting to attend the presentations above and visit the company at Exhibit Booth 2223. Blue Earth Diagnostics is hosting an Industry-Expert Theater event, “POSLUMA: Precision PET Imaging that is Truly Revealing,” with invited speaker Dr. Nicholas Zouain, Radiation Oncologist, Medical Director of West Florida Radiation Therapy, US Oncology Network, Clearwater, Fla. The event will be held on Sunday, October 1, 2023, from 12:00 PM to 1:00 PM PT, in Theater 1 of the San Diego Convention Center. For full session details and scientific presentation listings, please see the ASTRO online program here.
Indication and Important Safety Information About POSLUMA
INDICATION
POSLUMA (flotufolastat F 18) injection is indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer
- with suspected metastasis who are candidates for initial definitive therapy
- with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level
IMPORTANT SAFETY INFORMATION
- Image interpretation errors can occur with POSLUMA PET. A negative image does not rule out the presence of prostate cancer and a positive image does not confirm the presence of prostate cancer. The performance of POSLUMA for imaging metastatic pelvic lymph nodes in patients prior to initial definitive therapy seems to be affected by serum PSA levels and risk grouping. The performance of POSLUMA for imaging patients with biochemical evidence of recurrence of prostate cancer seems to be affected by serum PSA levels. Flotufolastat F 18 uptake is not specific for prostate cancer and may occur in other types of cancer, in non-malignant processes, and in normal tissues. Clinical correlation, which may include histopathological evaluation, is recommended.
- Risk of Image Misinterpretation in Patients with Suspected Prostate Cancer Recurrence: The interpretation of POSLUMA PET may differ depending on imaging readers, particularly in the prostate/prostate bed region. Because of the associated risk of false positive interpretation, consider multidisciplinary consultation and histopathological confirmation when clinical decision-making hinges on flotufolastat F 18 uptake only in the prostate/prostate bed region or only on uptake interpreted as borderline.
- POSLUMA use contributes to a patient’s overall long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk for cancer. Advise patients to hydrate before and after administration and to void frequently after administration. Ensure safe handling to minimize radiation exposure to the patient and health care providers.
- The adverse reactions reported in ≥0.4% of patients in clinical studies were diarrhea, blood pressure increase and injection site pain.
- Drug Interactions: androgen deprivation therapy (ADT) and other therapies targeting the androgen pathway, such as androgen receptor antagonists, may result in changes in uptake of flotufolastat F 18 in prostate cancer. The effect of these therapies on performance of POSLUMA PET has not been established.
To report suspected adverse reactions to POSLUMA, call 1-844-POSLUMA (1-844-767-5862) or contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Full POSLUMA prescribing information is available at www.posluma.com/prescribing-information.pdf.
For more information: www.blueearthdiagnostics.com
Find more ASTR23 coverage here
Related Prostate Cancer Content:
Blue Earth Diagnostics Announces Key Results from Phase 3 SPOTLIGHT Study of 18F-rhPSMA-7.3


 May 04, 2026
May 04, 2026 









