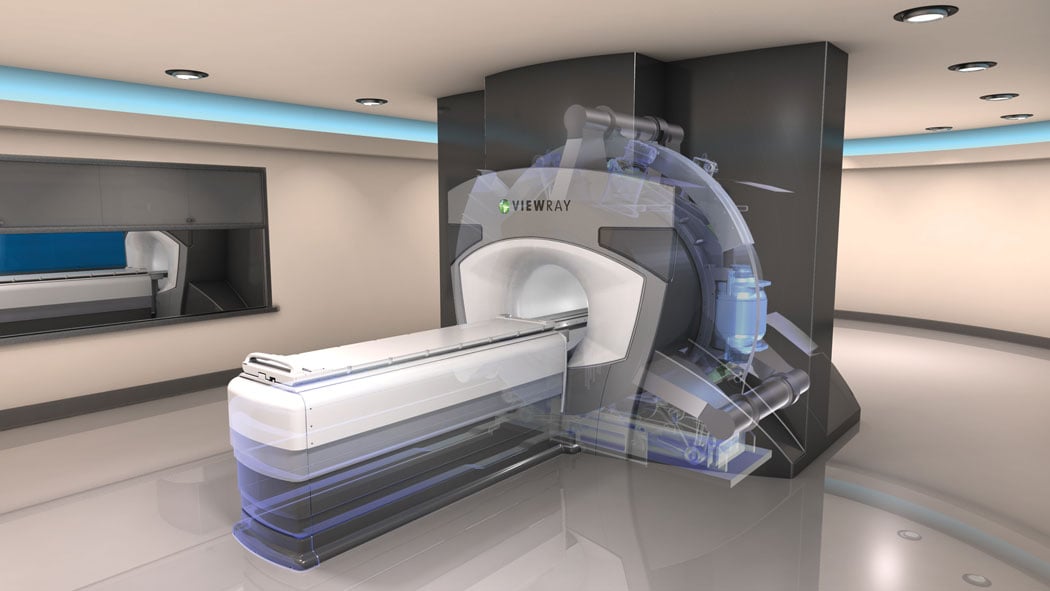
ViewRays MRIdian system directly tracks tumor motion continuously during treatment.
Image-guided radiation therapy (IGRT) affords greater accuracy of dose distribution during cancer treatment, allowing the radiation oncologist to see how a tumor is responding over the course of treatment. This has traditionally been accomplished with computed tomography (CT) or X-ray scans, which requires extra radiation exposure for the patient, with relatively poor contrast in soft tissue due to uniform electron density. Since treatment is only as good as the images provided, efforts are under way to find a better modality - and magnetic resonance imaging (MRI) may hold the answer.
Benefits of MRI
MRI-guided radiotherapy directly solves both of these problems, requiring no radiation for imaging while providing much greater viewing contrast in soft tissue - especially beneficial for imaging of the pelvic region.
Beyond these solutions, the technique provides a number of other benefits. Because there is no additional radiation dose to the patient, radiation oncologists can image more frequently, allowing real time viewing of treatment progression. In contrast, cone beam CT imaging is done in larger time intervals, so by the time treatment resumes, the images could be a few days old. This capability also makes it easier to track moving tumors in areas such as the lungs and stomach. Taken together, these allow radiation oncologists to practice truly adaptive radiation therapy for the first time.
"We can only treat what we see at the time of the treatment," said Prof. Uwe Oelfke, MCCPM, FInstP, head of the Joint Department of Physics at the Institute of Cancer Research (ICR) in London. "And if you don't see what you treat ... then you are shooting into the dark, basically, because we're trying to hit an invisible target with an invisible beam."
Taking the Linear Path
While MRI would seem to have numerous advantages over CT for image-guided radiotherapy - and those advantages seem to be understood in the radiology and radiation oncology communities - the technology has really only come into development in the last decade, with one company offering a system commercially to date.
The difficulty, largely, has been getting the two technologies - MRI scanners and the linear accelerators most often used in radiation therapy - to interact successfully without negating each other. In simple terms, the photons used during radiation therapy treatment generate their own electrons, whose path can be impacted by the magnetic field of the MRI scanner. This reaction can also occur within the soft tissue, and while not harmful can negate the effect of the treatment. Getting around this problem has been the main focus of those developing the technology, and several solutions have been found.
Elekta and Philips have been working to create an MRI-guided radiotherapy system that can still utilize a linear accelerator, since it is the current standard of care for radiation therapy. In 2012, the two companies formed the Elekta MR Linac Research Consortium, an international research initiative to develop a working MRI-guided radiotherapy system for clinical applications. The consortium includes an international cadre of research institutions, including Oelfke's team at the ICR in London, plus:
- Froedtert & the Medical College of Wisconsin Cancer Center (Milwaukee);
- MD Anderson Cancer Center (Houston);
- Sunnybrook Health Sciences Centre (Toronto);
- The Netherlands Cancer Institute-Antoni van Leeuwenhoek Hospital (Amsterdam);
- University Medical Center Utrecht (Utrecht, The Netherlands);
- Christie National Health System (NHS) Foundation Trust (Manchester, England); and
- The Royal Marsden NHS Foundation Trust (London).
Each institution is conducting its own research projects related to MRI-guided radiotherapy, with smaller cross-institutional working groups focusing on technical aspects - pulse sequences, workflow, appropriate imaging sequences, etc. - and potential applications - i.e., anatomies best suited for MRI radiotherapy. At press time, there are two operating systems, both at the UMC Utrecht (one was an initial experimental version and the second is a pre-production model) using a 1.5T magnetic field strength. Eventually each institution will have its own system, according to Jill Stief, director of product development, oncology systems for Elekta.
As the initiative progresses, Elekta and Philips will incorporate the sum total of all research findings into development of their commercial system, dubbed the Atlantic. On the current course, the partnership hopes to gain U.S. Food and Drug Administration (FDA) and CE mark approval in 2017 and begin shipping the first units in the United States and Europe in 2018. The Atlantic will employ Elekta's Monaco treatment planning system, including the Monte Carlo dose algorithm for more precise dose calculations.
Getting the linear accelerator and the MRI scanner to cooperate has proven successful thus far through two major modifications to the MRI scanner. The key, said Greg Trausch, senior product manager for Elekta, has been reducing the strength of the magnetic field where the linac is positioned, thereby improving the strength of the photon beam. Philips also designed new coils for the scanner that are resistant to the photon beam and allow more space for it to pass through. To further enhance performance, Elekta redesigned the gantry on the linear accelerator so that it is actually outside the treatment room and therefore cannot disrupt the magnetic field.
"It's not just about technology for the sake of technology," said Stief. "It's about the clinical practice and techniques that customers would use it for."
Cobalt Offers Alternatives
When the Atlantic enters the market in a few years, it will join ViewRay's MRIdian system, which debuted in May 2012 following FDA approval. MRIdian eliminates the linear accelerator from the equation, opting instead for three Cobalt-60 teletherapy heads. Cobalt generates gamma rays, which offer many of the same benefits as X-ray photons without the problems with the magnetic field.
ViewRay also made adjustments on the other side of the equation, using a 0.35T field strength magnet in the MRI scanner. This is significantly lower strength than most MRI scanners on the market, which are usually 1.5T or 3.0T. "The lower field strength avoids distortion to the radiation dose, while allowing for continuous imaging," said ViewRay CEO Chris Raanes.
The Future in Action
ViewRay has made three installs at present, all three of which are in the U.S. The first patients were treated at Siteman Cancer Center at Barnes-Jewish Hospital at Washington University in St. Louis in January 2014; the other two systems are at the University of Wisconsin Carbone Cancer Center and the University of California Los Angeles' (UCLA) Jonsson Comprehensive Cancer Center.
"We've been treating pretty much every part of the body," said Jeffrey Bradley, M.D., director of the S. Lee Kling Center for Proton Therapy at Barnes-Jewish Hospital at Washington University School of Medicine in St. Louis. Bradley's team has been using MRIdian to deliver both fractionated and stereotactic radiotherapy, with the potential to perform intensity-modulated radiation therapy (IMRT) as well. Bradley estimated the center has treated 150 patients since bringing MRIdian online. Adaptive treatment usually lasts roughly one hour and non-adaptive treatment takes about 30 minutes.
While treating patients, Siteman Cancer Center is also conducting a pair of clinical trials to further evaluate the technology. The first trial is studying the use of adaptive fractionated radiotherapy followed by a stereotactic boost for patients with lymph node-positive lung cancer; the second trial is focusing on stereotactic therapy for patients with a limited number of metastases who are also receiving chemotherapy and systemic treatment. Both trials are about halfway through their accrual period, according to Bradley.
"We think it'll change radiation therapy," he said of the technology.
These clinical trials are just the tip of the proverbial iceberg of what could be possible with MRI-guided radiation therapy, according to Raanes, who added that other MRIdian users are currently exploring the technology's ability to treat breast and liver cancer. "With breast cancer especially, it's nearly impossible to see the tumor bed on X-ray or CT, which is why many institutions do whole-breast irradiation therapy," he said, "But with MRI you can see much better, so you don't have to irradiate the entire breast."
One day, Oelfke believes it may even be possible to use MRI scans to assess the biological processes associated with tumor growth, identifying areas of greatest growth or resistance to combat cancer on a whole other level. "So after we have gained enough experience to control the anatomy, we can look inside the tumor and say, "˜Well, it's not the whole prostate; there's a dominant lesion in there,'" he said, "and we know exactly at the time of treatment where it is and we can give it the appropriate dose that it requires."
Related Articles on MRI-guided Radiation Therapy:
MRI-guided Radiation Therapy (2017)
First Patients Treated with ViewRay's MRIdian Linac at Henry Ford Health System
Early Clinical Experience with ViewRay's MRIdian Linac Presented at AAPM 2017
Elekta Begins MR-Linac Installation at Sunnybrook Health Sciences Centre


 April 30, 2026
April 30, 2026 









