Calgary Scientific Inc. announced it has become a member of the Health and Life Sciences Technology Alliance Partner (TAP) program of Hitachi Data Systems and will partner with Hitachi Data Systems to deliver the next generation of vendor neutral archive (VNA) solutions built on the Hitachi Clinical Repository platform.
EIZO offers the most extensive line-up of high quality LCD monitors into the medical industry. Focusing on monitors that ...
The Radiation Oncology Institute (ROI) has selected Malolan S. Rajagopalan, M.D., radiation oncology resident, University of Pittsburgh Cancer Institute, to receive a $20,000 grant for a project to compile best practices regarding the management of radiation therapy toxicity.
AT A GLANCE Organization: Expert Radiology Management Services, LLC Specialty: Subspecialty teleradiology — neuro and ...
Southco Inc., an engineering solutions company, has expanded its T Series tilt display mount product line, offering versions with or without counterbalanced constant torque.
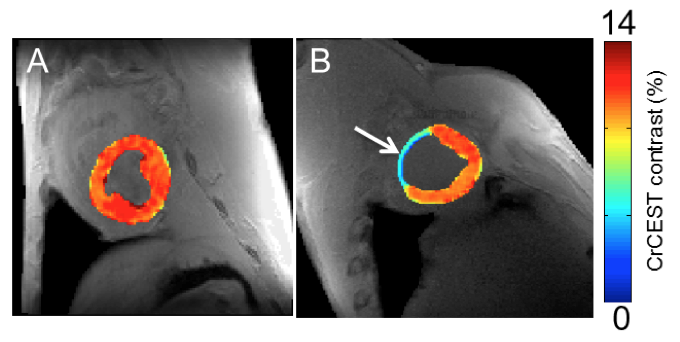
A new magnetic resonance imaging (MRI) method to map creatine at higher resolutions in the heart may help clinicians and scientists find abnormalities and disorders earlier than traditional diagnostic methods.
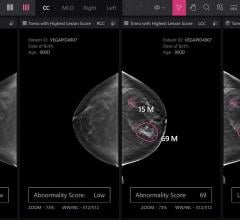
Matakina International announced that The Hospital of Central Connecticut (HOCC) is the first facility in Connecticut to adopt VolparaDensity breast imaging software, designed to help overcome the limitations of mammography to detect cancer in women with dense breasts. The Enterprise installation of HOCC provides the ability to offer objective volumetric breast density values for mammograms performed at six facilities on eight Hologic full-field mammography systems.
Radiology departments have many different needs and face a wide variety of challenges that can impact their departments ...
Radcal Corp. announced partnership with its French distributor, Medi Test, which opened up its own certified calibration and repair facility, Medix Lab, June 2013.
Barco has teamed up with Mammography Education Inc. to supply its latest mammography display systems and dedicated support for hands-on mammography CME courses designed to teach radiologists how to detect early-phase breast cancers.
Patients with the most common form of focal epilepsy have widespread, abnormal connections in their brains that could provide clues toward diagnosis and treatment, according to a new study published online in the journal Radiology.
Despite decades of progress in breast imaging, one challenge continues to test even the most skilled radiologists ...
David Widmann, president and COO of Konica Minolta Medical Imaging, discusses the company's commitment to primary ...
IBA Dosimetry is excited to launch its new Digital Imaging Analysis Software IQ Analyzer Primus at the RSNA 2013. Image ...
AirXpanders Inc. announced new data published in The Journal of Radiation Oncology that demonstrates the AeroForm Tissue Expander can withstand radiation levels well above the maximum dose level typically used in post-mastectomy radiation therapy.
Bayer Radiology’s Barbara Ruhland and Thom Kinst discuss how radiology departments can address the many different ...
With a commitment to delivering the right dose, a desire to keep patient satisfaction high, and the challenge of ...
Swissray International Inc., a digital radiography (DR) vendor, said it completed the acquisition of the Norland assets from Cooper Surgical Inc.
Primordial announced the addition of numerous customers, its entrance into the radiologist reporting system market and the development of new capabilities across all of its product lines.
Viztek, a provider of digital software and hardware diagnostic imaging solutions, announced that the Platinum Dynamic Fluoroscopy System has received U.S. Food and Drug Administration (FDA) 510(k) clearance.
A clinical trial for breast cancer, published in the medical journal The Lancet, shows that a single fraction of targeted intraoperative radiotherapy (TARGIT) delivered with the Zeiss Intrabeam at time of lumpectomy is non-inferior to traditional external beam radiation (EBRT) delivered over three to six weeks after breast conserving surgery, for a select group of patients.
Modus Medical Devices Inc. debuted Quasar MRID3D, a patent-pending method for quantifying geometric distortion in 3-D magnetic resonance (MR) images, at the Radiological Society of North America Annual Meeting (RSNA 2013) in Chicago.
ClearCanvas Inc., developer, manufacturer and supplier of imaging informatics infrastructure solutions, highlighted two informatics solutions at the Radiological Society of North America Annual Meeting (RSNA 2013) in Chicago.
Dilon Technologies Inc. announced that it acquired the assets of the Navigator gamma probe business from RMD Instruments Corp., manufacturer and marketer of intraoperative surgical probes and subsidiary of Dynasil Corporation of America.

 January 16, 2014
January 16, 2014