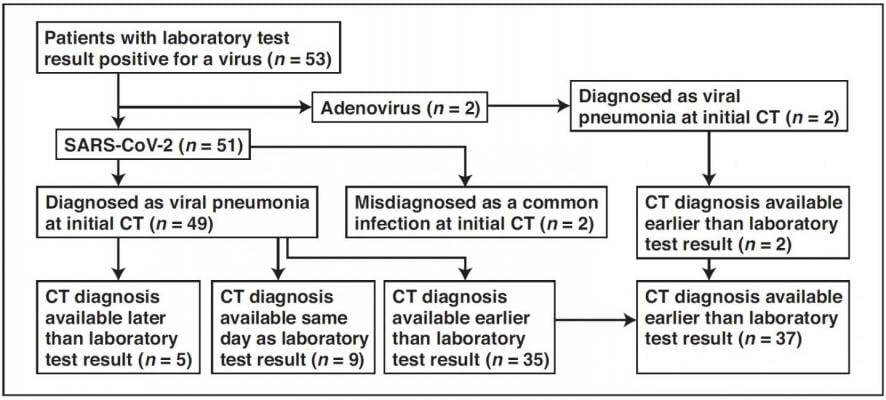
Flowchart shows time difference between positive laboratory test results and positive CT findings for COVID-19 and adenovirus infection in the study group. Image courtesy of American Journal of Roentgenology (AJR)
March 6, 2020 — An article by radiologists from Wuhan, China — published open-access and ahead-of-print in the American Journal of Roentgenology (AJR) — concluded that chest CT had a low rate of misdiagnosis of COVID-19 (3.9 percent, 2/51) and could help standardize imaging features and rules of transformation for rapid diagnosis; however, CT remains limited for the identification of specific viruses and distinguishing between viruses.
Yan Li and Liming Xia at Tongji Hospital in Hubai Province studied the first 51 patients diagnosed with COVID-19 infection confirmed by nucleic acid testing (23 women and 28 men; age range, 26-83 years) and two patients with adenovirus (one woman and one man; ages, 58 and 66 years). In their retrospective cohort of 53 patients, as of February 9, a total of 99 chest CT examinations had been performed.
Comparing image reports of the initial CT study with laboratory test results to identify patterns suggestive of viral infection, according to Li and Xia, "COVID-19 was misdiagnosed as a common infection at the initial CT study in two patients with underlying disease and COVID-19."
Meanwhile, viral pneumonia was correctly diagnosed at the initial CT study in the remaining 49 patients with COVID-19 and two patients with adenovirus.
As Li and Xia explained: "CT of one of the two patients with confirmed adenovirus infection showed ill-defined patchy ground-glass opacities (GGOs), segmental and subpleural consolidations in both lungs, and pleural effusion. CT of the other patient showed subpleural GGOs and consolidation with vascular enlargement, interlobular septal thickening, and air bronchogram sign."
The CT findings seen in Li and Xia's two adenovirus cases were similar to those observed in their COVID-19 cases.
The two authors also found CT features of COVID-19 that differ from both severe acute respiratory syndrome coronavirus (SARS-CoV) and Middle East respiratory syndrome coronavirus (MERS-CoV): a reversed halo sign in two patients (3.9 percent) and pulmonary nodules with a halo sign in nine patients (17.6 percent).
"These findings are not mentioned, to our knowledge, in the studies in the literature," the authors noted.
"It is valuable for radiologists to recognize that the CT findings of COVID-19 overlap with the CT findings of diseases caused by viruses from a different family, such as adenovirus, and have differences as well as similarities with viruses within the same family, such as SARS-CoV and MERS-CoV," added Li and Xia.
For more information: www.arrs.org
Additional COVID-19 Resources for Clinicians:
World Health Organization (WHO) COVID-19 situation reports
World Health Organization (WHO) coronavirus information page
U.S. Food and Drug Administration (FDA) COVID-19 information page
Centers for Disease Control (CDC) COVID-19 information page
Related Coronavirus Content:
The Cardiac Implications of Novel Coronavirus
CT Provides Best Diagnosis for Novel Coronavirus (COVID-19)
Radiology Lessons for Coronavirus From the SARS and MERS Epidemics
Deployment of Health IT in China’s Fight Against the COVID-19 Epidemic
Emerging Technologies Proving Value in Chinese Coronavirus Fight
Radiologists Describe Coronavirus CT Imaging Features
Coronavirus Update from the FDA
CT Imaging of the 2019 Novel Coronavirus (2019-nCoV) Pneumonia
CT Imaging Features of 2019 Novel Coronavirus (2019-nCoV)
Chest CT Findings of Patients Infected With Novel Coronavirus 2019-nCoV Pneumonia


 May 19, 2026
May 19, 2026 









