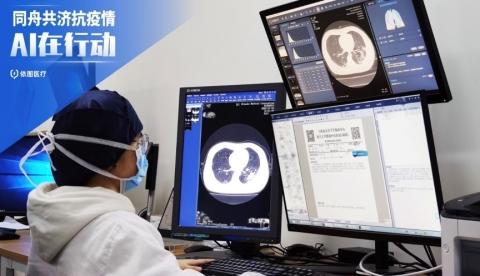
Doctor in our hospital is using this intelligent system for accurate diagnosis. (Photo: Business Wire)
March 31, 2020 — The Intelligent Evaluation System of chest computed tomography (CT) for COVID-19, developed by YITU under the guidance of Shanghai Public Health Clinical Center, was officially launched and was put into the first-line battle against COVID-19 pandemic on Jan. 28. It is the first AI-equipped diagnostic assistant, which significantly improves efficiency in identifying coronavirus through automatic detection and quantitative analysis within 2-3 seconds, and eases the burden of Chinese doctors.
YITU Healthcare reinforced its R&D capability with multi-departments cooperation right after the outbreak of the pandemic and released its cutting-edge AI diagnosis system, with upgrades on a daily basis during the Chinese New Year. YITU assisted many designated medical institutions and supported the frontline heroes fighting the pandemic.
According to its official public account of Shanghai Public Health Clinical Center, one of the authoritative institutions in the field of infectious diseases in China and the key unit for the treatment of COVID-19 in Shanghai, an article titled "AI Aids the Scientific Prevention and Control, Intelligent Evaluation System for COVID-19 Launched in Shanghai Public Health Clinical Center", announced its latest achievements in using artificial intelligence technology to help doctors carry out quantitative analysis and evaluation of therapeutic efficacy of new coronavirus lesions based on CT images.
COVID-19 Diagnosis and Treatment Plan (Trial Version 7) issued by the General Office of the National Health Commission of the People's Republic of China stipulates that CT imaging results should be used as one of the criteria for the diagnosis of COVID-19 nationwide. At the same time, based on clinical experience, the CT imaging performance of COVID-19 is further clarified and the quantitative evaluation of CT images is incorporated into the clinical classification basis.
YITU's AI-driven solution of intelligent diagnosis and quantitative evaluation of CT images of COVID-19, are through industry-leading image algorithms, and grade the severity of various pneumonia diseases of local lesions, diffuse lesions, and whole lung involvement. Furthermore, it accurately quantifies the cumulative pneumonia load of the disease through quantitative and omics analysis of key image features such as the morphology, range, and density of the lesion, and achieves a dynamic 4-D contrast of the lung lesions on the CT, helping in clinical judgment of the condition, evaluation of the efficacy, and prediction of the prognosis.
Taking up the challenge and responsibility in response to frontline needs and requests, YITU's Intelligent Evaluation System of Chest CT for COVID-19 has been adopted across 25 provinces and municipal autonomous prefectures in 100 medical institutions and served over 100,000 person-times in China by far.
For more information: www.yitutech.com
Related Coronavirus Content:
VIDEO: Use of Telemedicine in Medical Imaging During COVID-19
VIDEO: How China Leveraged Health IT to Combat COVID-19
CDRH Issues Letter to Industry on COVID-19
Qure.ai Launches Solutions to Help Tackle COVID19
ASRT Deploys COVID-19 Resources for Educational Programs
Study Looks at CT Findings of COVID-19 Through Recovery
VIDEO: Imaging COVID-19 With Point-of-Care Ultrasound (POCUS)
The Cardiac Implications of Novel Coronavirus
CT Provides Best Diagnosis for Novel Coronavirus (COVID-19)
Radiology Lessons for Coronavirus From the SARS and MERS Epidemics
Deployment of Health IT in China’s Fight Against the COVID-19 Epidemic
Emerging Technologies Proving Value in Chinese Coronavirus Fight
Radiologists Describe Coronavirus CT Imaging Features
Coronavirus Update from the FDA
CT Imaging of the 2019 Novel Coronavirus (2019-nCoV) Pneumonia
CT Imaging Features of 2019 Novel Coronavirus (2019-nCoV)
Chest CT Findings of Patients Infected With Novel Coronavirus 2019-nCoV Pneumonia


 May 22, 2026
May 22, 2026 









