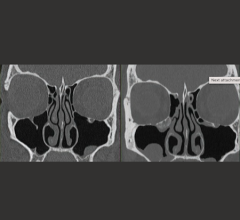
Medic Vision, a pioneer and US market leader in 3rd party XR-29 standard solutions, announced July 19 that it was granted a patent by the U.S. Patent and Trademark Office on its XR-29 Dose-Check system (Patent No. 9693747), which is an integral part of the notable market-leading SafeCT-29 product suite.
The SafeCT-29 product suite, which includes the XR-29 Dose Check component, was the first and only third-party solution to receive FDA clearance, and remains the only third-party solution that has been endorsed by the OEMs. Since being officially launched, SafeCT-29 has been installed in hundreds of medical care centers throughout the US, making Medic Vision the leading NEMA XR-29 third-party compliance solution provider.
“We had put a lot of time and effort into the development of SafeCT-29, and are extremely proud of the innovative solution and technology we created to address the NEMA XR-29 Dose Check requirements.” said CEO Eyal Aharon of Medic Vision. “Advantageously, adding SafeCT-29 with our Dose Check feature to existing CT systems provides compliance with the most challenging aspects of the XR-29 standard without requiring hospitals and imaging centers to purchase new CT systems.”
Medic Vision’s market position is now likely to be significantly enhanced by virtue of the issuance of its latest patent and the company stated that it fully expects providers of medical imaging services and solutions to respect Medic Vision’s patent rights.
For more information: www.medicvision.com/usa/products/

 February 04, 2026
February 04, 2026