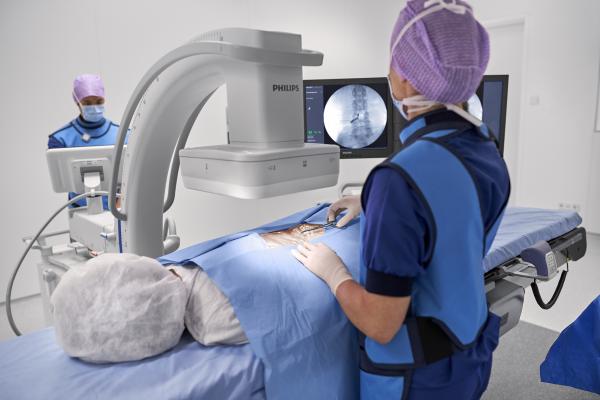
February 18, 2019 — Philips announced the launch of Philips Zenition, its new mobile C-arm imaging platform. Mobile C-arms are X-ray systems that are brought into the operating room (OR) to provide live image guidance during a wide range of surgeries including orthopedic, trauma and vascular procedures. The Zenition mobile C-arm platform brings together innovations in image capture, image processing, ease-of-use and versatility pioneered on Philips’ Azurion platform. Zenition allows hospitals to maximize OR performance, enhance their clinical capabilities and offer their staff a high-quality user experience. Zenition will be introduced in the U.S., Germany, Austria and Switzerland in the first half of 2019, and will subsequently be rolled out in further markets.
The scope and complexity of surgical interventions – especially in the rapidly growing area of image-guided minimally invasive surgery – continues to increase, as does the number of patients requiring treatment. In order to treat more patients at a lower cost, hospitals require a versatile fleet of C-arms with varying capabilities that easily adapt to the needs of different types of surgery and different operators. Zenition mobile C-arms are easy to move between operating rooms, simple to position around the patient and intuitive to operate.
Read the article "Mobile C-Arms Mature But Improving"
“The Philips Zenition is a user-friendly system that’s intuitive to use for both surgeons and nursing staff,” said Nikolaos Bonaros, associate professor of cardiac surgery at the Medical University of Innsbruck, Austria. “Its simplified workflow means that we can convert a room from a conventional OR to a high-quality interventional room more quickly. At the same time the system provides high image quality at the level required for hybrid OR procedures.”
Philips Zenition supports increased OR performance across the healthcare facility. The platform’s tablet-like user interface and simple Unify workflow mean that once an operator has learned to use one system on the platform, it is easy for them to operate them all. The systems make point-and-shoot image capture fast and intuitive during any interventional or surgical procedure. Zenition’s compact design, Position Memory feature (only available on Zenition 70) and BodySmart software, which captures fast and consistent images even at the edge of the image intensifier or flat detector, reduce the need for C-arm repositioning by 45 percent [1].
For crisp high-quality image viewing, Zenition C-arm systems incorporate the same image processing algorithms used on the company’s Azurion platform, offering high-definition visualization of patient anatomies and a greater viewing area. Image quality is assured by features such as Philips’ MetalSmart software, which automatically adjusts the contrast and brightness of images to improve image quality when metal objects such as implants are present in the field of view – a feature that makes Zenition systems especially useful in orthopedics.
The Philips Zenition platform is designed for the future, allowing for longer clinical relevance of the systems. With two different detector and image intensifier options and the ability to subscribe to Philips’ Technology Maximizer program, Zenition offers users the latest software and hardware technology releases for a fraction of the cost of purchasing them individually.
Philips’ Zenition C-arms are CE marked and have received 510(k) clearance from the U.S. Food and Drug Administration (FDA). They will be showcased at the 2019 European Congress of Radiology (ECR) exhibition, Feb. 28 – March 3 in Vienna, Austria.
Read the article "Mobile C-Arms Going Digital"
For more information: www.usa.philips.com/healthcare
Reference
[1] Results obtained during user tests performed in November 2013 by Use-Lab GmbH, an independent company. The tests involved 30 U.S.-based clinicians (15 physicians teamed up with 15 nurses or X-ray technicians), who performed simulated procedures in a simulated OR environment. None of them had worked with a Philips C-arm or with each other before.


 April 15, 2026
April 15, 2026