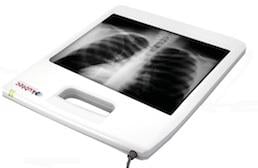
October 25, 2011 — Kubtec, a manufacturer of digital specimen X-ray systems, has received FDA approval on its Digiview 250. Designed for portable imaging applications and positionable with one hand, the Digiview 250 is a tool for neonatal imaging, as well as the examination of extremities, for use in mobile systems and general radiology applications.
Packaged in a durable, custom-molded plastic housing with an integrated handle, the Digiview 250 is the first portable CMOS X-ray panel system of its size with a weight under 7 pounds. Additionally, at 96µh pixels, the Digiview 250 provides the highest resolution images in the industry. The panel’s active-pixel CMOS photodiode array consists of more than 5 million individual pixels, while the low-noise, 14-bit digital output produces crisp, sharp images. Fiber-optic technology increases sensor lifetime and enhances detective quantum efficiency at energies up to 160 kVp. The highly sensitive detector acquires images at 50% lower radiation levels than competitive systems, Kubtec says.
The Digiview 250 features Kubtec’s DICOM 3.0-compliant Digicom software, which provides rapid image acquisition and access to a full suite of user-friendly image enhancement tools. The suite incorporates Kubtec’s exclusive customizable filters, 3-D and measurement tools and the ability to annotate and send multiple images to picture archiving and communications systems (PACS). With a cable tether, the Digiview 250 can be connected to a laptop or PC via standard gigabit Ethernet or fiber-optic interface. The Digiview 250 requires less than 10W of power and runs off a standard AC power adapter or battery pack.
The Digiview will be shown in Kubtec’s booth #5517 at the Radiological Society of North America’s (RSNA) annual meeting in Chicago, November 27 – December 4, 2011.
For more information: www.kubtec.com


 June 03, 2026
June 03, 2026 









