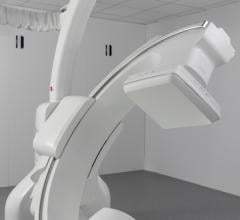
MobileDaRt Evolution MX9 Version (Photo: Shimadzu Medical Systems USA)
Nov. 21, 2025 — Shimadzu Medical Systems USA (SMS), a division of Shimadzu Precision Instruments, Inc. (SPI) which is fully owned by Shimadzu Corp., Japan, has announced the release of the MobileDaRt Evolution MX9 Version.
Mobile X-ray systems enable radiography to be performed in patient rooms, emergency rooms, operating rooms or at any location in hospitals where an X-ray examination is required. Shimadzu's mobile X-ray system provides intuitive maneuverability and great forward visibility during travel. The acquired image can be verified on the built-in monitor immediately to assist operator's quick decision for treatment.
Since the system is used by a multitude of healthcare providers, there has been an increase in demand for streamlined operations that deliver simplified workflow and reduce the frequency of retakes. Responding to the needs for the clinical environment, Shimadzu offers VISION SUPPORT, an imaging support function utilizing camera applications. Also, the MX9 includes a tube-mounted monitor. This provides a secondary workspace, making it possible to enhance workflow efficiency and provide essential information for dose management.
To meet the diverse needs of medical facilities, the MobileDaRt Evolution supports three types of DR systems, each featuring advanced flat panel detectors (FPDs) and integrated digital technology. The latest MobileDaRt Evolution MX9 Version c type enhances workflow efficiency with a secondary monitor that conveniently displays patient information, live camera images, and protocol selections right at your fingertips.
This product will be showcased at the RSNA (Radiological Society of North America) Technical Exhibits, in Chicago.
For more details, visit
Mobile X-ray : SHIMADZU MEDICAL SYSTEMS USA


 March 31, 2026
March 31, 2026