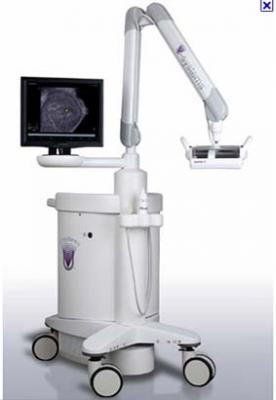
May 19, 2011 - U-Systems launched a new transducer technology designed to deliver unparalleled imaging performance and patient comfort. Reverse-Curve Crystal (RCC) Transducer Technology was designed and optimized for use with the U-Systems’ somo•v Automated Breast Ultrasound (ABUS) system. The announcement was made at the 10th Post Graduate Course of the Society of Breast Imaging (SBI) Meeting, in San Antonio, May 18- 21.
As part of the launch, U-Systems introduced the C14-6XW (CXW) ultra-broadband transducer featuring Reverse-Curve Crystal technology. The ergonomic RCC design conforms to anatomic curve of women’s breasts for improved comfort and imaging performance during an automated 3-D ultrasound exam. RCC technology enables convergent scan line geometry, which allows ultrasound beams, or sound waves, to penetrate the skin perpendicularly, minimizing beam refraction, improving penetration and sharpening focus.
Additionally, the CXW transducer creates uniform compression thickness across the entire breast. Greater image overlap ensures entire field-of-view imaging, increasing confidence of viewing entire breast and minimizing refraction artifacts at tissue interface.
“The CWX Reverse-Curve transducer matches the natural curve of women’s breasts, providing greater tissue coverage, resulting in fewer contact artifacts and greater patient comfort,” said Susan G. Roux, M.D., medical director of the Community Hospital of the Monterey Peninsula’s Breast Care Center. “Better coverage, fewer artifacts, plus the improved detail resolution not only gives me greater confidence I’m viewing the entirety of dense breast tissue, it also increases my overall diagnostic confidence.”
U-Systems also introduced its new Soft-Touch Floating Compression Membrane. Designed specifically for use with the somo•v ABUS System, the Soft-Touch Membrane provides the highest exam quality with a greater level of patient comfort. It is designed to instantly adapt to woman’s unique anatomy and evenly distribute pressure across the breast for a quick, comfortable exam. The anatomic shape will also expedite patient positioning for improved workflow.
In addition to the RCC transducer technology, the company also showcased ABUS Scan Station and Workstation software improvements at the SBI meeting. Developed to help radiologists simplify their reading protocol and reach higher levels of productivity, Release One•6 features robust software improvements enabling easy and rapid access to prior exams and images, faster exam loading and customizable user- and session-specific protocols for improved workflow and batch reading efficiency.
The somo•v ABUS incorporates automated ultrasound technology FDA-cleared for adjunctive diagnostic use with mammography, the somo•v ABUS was developed as the foundation technology for U-Systems’ ground-breaking, prospective SOMO•INSIGHT clinical study, the largest clinical study ever conducted by an ultrasound company.
The study is designed to evaluate whether ABUS, in combination with digital mammography, is more sensitive than a routine screening mammogram alone in detecting breast cancer in women with dense breast tissue. More than 15,000 women have participated in the study, which is actively recruiting up to 20,000 women at multiple breast imaging centers nationwide. To date, the study has identified a significant number of cancers missed by mammography and detected by ABUS.
For more information: www.u-systems.com.


 May 07, 2026
May 07, 2026 









