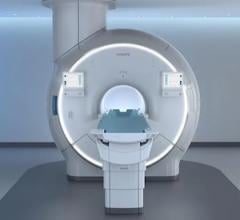
January 25, 2012 — InSightec Ltd. announced that it has submitted a premarket approval application (PMA) to the U.S. Food and Drug Administration (FDA) for its ExAblate treatment of pain palliation of bone metastases. ExAblate uses magnetic resonance guided focused ultrasound (MRgFUS) to treat the pain caused by bone metastases non-invasively and without ionizing radiation.
Over 60 percent of cancer patients suffer from painful bone metastases. Of these, 30 percent will not respond to radiation therapy. ExAblate was investigated as an option for these patients. The PMA submission was based on a two-arm randomized controlled trial comparing patients undergoing ExAblate's MR guided focused ultrasound for palliation of painful bone metastases with patients undergoing no treatment.
Patients who had no response to the sham treatment were later allowed to undergo an ExAblate treatment. The FDA also granted an expedited review process for the PMA submission.
ExAblate combines high intensity focused ultrasound to thermally ablate tumors and continuous MRI to visualize the anatomy, plan treatment and monitor treatment outcomes in real time.
ExAblate is the only MRgFUS system with FDA approval, granted in 2004 for uterine fibroids. It also received European CE marking for uterine fibroids, bone metastases, and adenomyosis. Clinical trials are ongoing for brain disorders, including essential tremor and Parkinson's disease, and prostate cancer.
For more information: www.insightec.com, www.bone-pain-palliation.co.uk/


 May 22, 2026
May 22, 2026