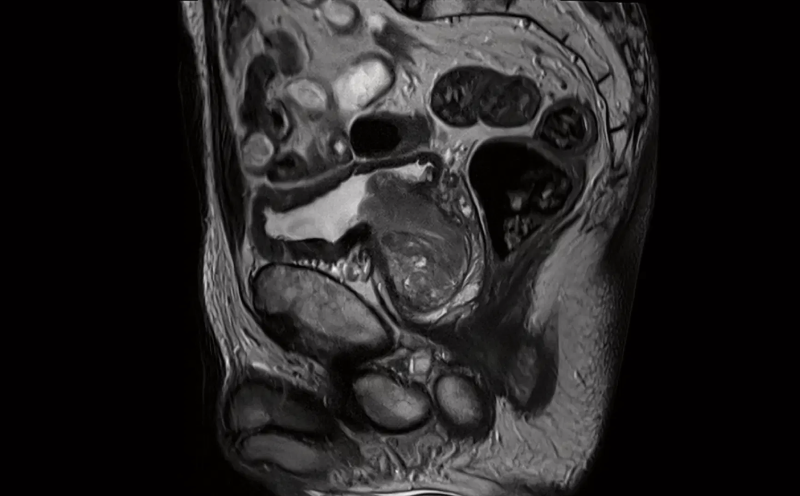
October 31, 2023 — Prostate cancer has a significant impact globally. For example, approximately one in eight men in the ...
Magnetic Resonance Imaging (MRI)
MRI creates images from the magnetic resonance created in hydrogen atoms when they are polarized and an electromagnetic pulse is used to knock them off axis. This section includes MR analysis software, MRI scanners, gadolinium contrast agents and related magnetic resonance imaging accessories.
October 25, 2023 — Esaote, a leading Italian company in ultrasound, dedicated MRI and healthcare IT, presents S-scan ...
October 20, 2023 — In certain cases, a new method can provide as much information from brain images taken with computed ...
Fujifilm’s APERTO Lucent is a 0.4T mid-field, open MRI system addressing today’s capability and image quality needs ...
October 19, 2023 — Alexandra Touroutoglou, PhD, and Bradford Dickerson, MD, neuroscientists in the department of ...
October 17, 2023 — Halo Precision Diagnostics, a leader in early disease detection using precision diagnostics, today ...
October 12, 2023 — Hyperfine, Inc., a groundbreaking medical device company that has redefined brain imaging with the ...
In June, the Philips Radiology Experience Tour hit the road to provide healthcare professionals with an opportunity to ...
October 5, 2023 — A new study, led by radiation oncology physicists at Miami Cancer Institute, part of Baptist Health ...
September 28, 2023 — Siemens Healthineers has announced the Food and Drug Administration (FDA) clearance of the Magnetom ...
Since the advent of the magnetic resonance imaging (MRI) exam on human patients in the late 1970s, this innovation ...
This summer, the Philips Radiology Experience Tour has been bringing Philips imaging modalities directly to the ...
September 22, 2023 — According to an accepted manuscript published in the American Journal of Roentgenology (AJR) ...
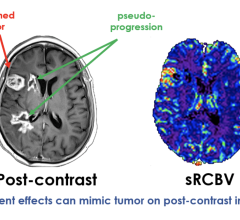
September 21, 2023 — Imaging Biometrics, LLC announced the results of a study that validates IB Neuro’s processing of ...
In June, the Philips Radiology Experience Tour hit the road to provide healthcare professionals with an opportunity to ...
The healthcare industry faces many different types of obstacles in today’s challenging marketplace. Staff shortages ...
The world of medical imaging is marking a significant milestone in 2023: the 50th anniversary of magnetic resonance ...
September 14, 2023 — Insight Medbotics, a medical device company combining the accuracy of MRI with the precision of ...
September 11, 2023 — Polarean Imaging plc (AIM: POLX), a commercial-stage medical device leader in advanced magnetic ...
September 8, 2023 — A simple, noninvasive contrast enhanced ultrasound (CEUS) scan is an ideal tool for resolving ...
According to a new report from Transparency Market Research (TMR), PET radiotracers are expected to rise at a CAGR of 8 ...
August 24, 2023 — Medical imaging via X-rays, CT scans, MRIs and ultrasounds provide health-care professionals with ...
August 23, 2023 — Fujifilm Healthcare Americas Corporation, a leading provider of diagnostic imaging and medical ...
August 22, 2023 — Using MRI as a screening test alongside PSA density allowed detection of cancers that would have been ...


 October 31, 2023
October 31, 2023