September 21, 2023 — Imaging Biometrics, LLC announced the results of a study that validates IB Neuro’s processing of reduced-dose gadolinium-based contrast agent (GBCA) perfusion studies on 1.5T scanners.
The multi-center study, led by Dr. Mark Shiroishi, MD, Assistant Professor of Neuroradiology and Director of Neuro-Oncology Imaging at the Keck School of Medicine of USC, is an extension of the foundational study published in April 2019 (Moving Towards a Consensus DSC-MRI Protocol: Validation of A Low Flip Angle Single Dose Option as a Reference Standard for Brain Tumors) that validated the use of a low-dose protocol for acquiring MRI perfusion data. In the 2019 study, it was shown that 3T MRI data collected with 50% less GBCA could provide perfusion maps comparable to the higher dose standard when processed using IB Neuro.
IB Neuro has now been validated using this approach on 1.5T scanners.
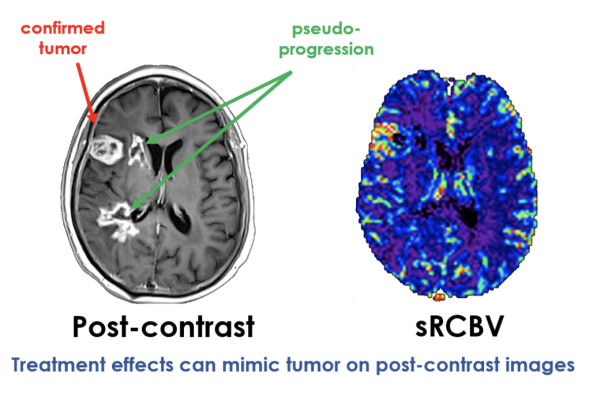
Dynamic susceptibility contrast (DSC) MRI is the most common approach used to measure brain tumor perfusion. Relative cerebral blood volume (rCBV) is the most widely generated parameter map that provides important information regarding tumor grade, prognosis, and treatment response. IB Neuro automatically processes DSC data using its unique contrast leakage (of GBCA) correction technology. It is the only platform that generates quantitative output that can be directly compared across timepoints. This study also makes IB Neuro the only platform proven to provide accurate perfusion maps under low-dose conditions at both 1.5T and 3T MR scanners.
“The study demonstrates that a single dose (no preload), low flip angle method does not compromise image quality,” said Dr. Mark Shiroishi, MD. “This is a great added benefit for patients who may have concerns about GBCA risks, and for administrators looking for opportunities to curtail operational expenses”.
For more information: https://www.imagingbiometrics.com/


 May 18, 2026
May 18, 2026 









