If you enjoy this content, please share it with a colleague
RELATED CONTENT
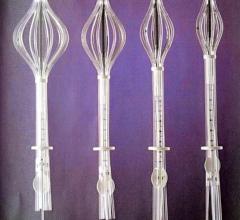
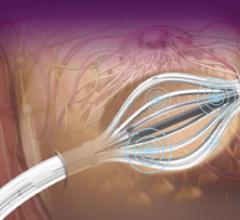
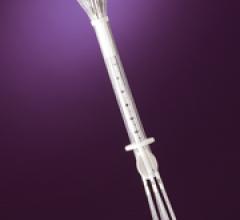
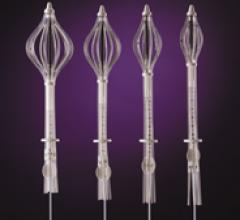
Cianna Medical Inc. is a women’s health company. It announced low recurrence rates – even for younger women – as part of a retrospective analysis of 673 patients with invasive breast cancer treated with the Savi breast brachytherapy applicator. Savi is a strut-based device that delivers breast brachytherapy, a 5-day course of targeted radiation for early-stage breast cancer.
Cianna Medical Inc., a women’s health company, conducted a large, retrospective study evaluating the safety and efficacy of the Savi breast brachytherapy applicator. Researchers reported favorably low recurrence rates, high rates of survival and excellent cosmetic outcomes. Savi is a strut-based applicator that delivers a form of accelerated partial breast irradiation (APBI), a five-day course of targeted radiation for early-stage breast cancer.
Cianna Medical, Inc., a women’s health company, today announced the outcomes of a large, retrospective study demonstrating the safety and efficacy of the SAVI breast brachytherapy applicator. Researchers reported favorably low recurrence rates, high rates of survival and excellent cosmetic outcomes. SAVI is a strut-based applicator that delivers a form of accelerated partial breast irradiation (APBI) known as breast brachytherapy, a 5-day course of targeted radiation for early-stage breast cancer.
Cianna Medical Inc. announced approval from the Japanese Ministry of Health, Labor and Welfare (MHLW) to market its strut adjusted volume implant (SAVI) applicator in Japan. SAVI delivers accelerated partial breast irradiation (APBI), a five-day course of targeted radiation for early-stage breast cancer patients. The company has partnered with MC Medical Inc., a subsidiary of Mitsubishi Corporation, to bring SAVI to the Japanese healthcare market.
Cianna Medical Inc., announced results of two new studies showing excellent longer-term outcomes with the SAVI breast brachytherapy applicator, including low recurrence rates, few toxicities, excellent cosmetic outcomes and high rates of survival. A third study demonstrated SAVI’s ability to precisely deliver radiation while avoiding critical structures. SAVI is a strut-based applicator that delivers accelerated partial breast irradiation (APBI), a five-day course of targeted radiation for early-stage breast cancer.
A potential paradigm shift is gathering momentum in the treatment of early-stage breast cancer, driven by data showing strong clinical outcomes for the use of more sophisticated therapies. Newer therapies using accelerated partial breast irradiation (APBI, also referred to as breast brachytherapy) not only allow women to remain whole, they also offer properly selected patients several advantages over traditional whole breast irradiation (WBI).
May 29, 2012 — Cianna Medical recently announced the launch of BEST (Brachytherapy Efficacy, Safety and Treatment) Forum, a new educational resource for physicians to obtain the latest research in breast brachytherapy and gain insights into best practices and available technology.
May 9, 2012 - The American Society of Breast Disease (ASBD) is concerned about misleading information in a recent study published in the Journal of the American Medical Association (JAMA), comparing breast brachytherapy to whole breast irradiation (WBI).
April 2, 2012 — Two new studies demonstrate the cosmetic and dosimetric advantages of the SAVI breast brachytherapy applicator for treatment of early-stage breast cancer. The studies were presented as scientific posters at national medical conferences and were drawn from 12 centers participating in the SAVI Collaborative Research Group.
March 15, 2012 - Two new studies demonstrate the cosmetic and dosimetric advantages of the SAVI breast brachytherapy applicator for treatment of early-stage breast cancer.


 March 11, 2014
March 11, 2014