April 1, 2009 – Siemens Healthcare is set to demonstrate the benefits of workflow automation on the new release of its premier general imaging platform, the ACUSON S2000 ultrasound system, at the annual conference of the American Institute of Ultrasound in Medicine (AIUM 2009), this week.
Siemens will also highlight how automation can support the viability of fusing ultrasound and computed tomography (CT) imaging in the clinical routine. Offering the best of both worlds, CT and ultrasound fusion combines the benefits of real-time ultrasound imaging with global imaging display of CT. However, current fusion techniques require bulky transmit and receive equipment to track the patient’s anatomy in real time. Also, patients need to lie completely still during the entire exam to prevent elaborate manual realignments before the examination can continue.
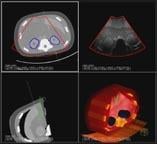
According to Siemens, the limitations of handheld ultrasound in terms of scan time and user-variability have limited ultrasound’s role in imaging to that of a secondary modality to look at a palpable mass or a finding identified on a mammogram. The ACUSON S2000 Automated Breast Volume Scanner (ABVS) has the potential to change that. In less than 15 minutes, the system acquires full-field sonographic volumes of the breast. These volumes offer new insight into the intricate anatomy and architecture of the breast, including the intuitive anatomical coronal view, which has not been available using conventional hand held ultrasound, according to the company.
After acquisition, the volume data is sent to a workstation for comprehensive offline review and diagnosis, as well as semi-automated reporting and BI-RADS reporting. A complete ultrasound breast imaging solution, the ACUSON S2000 ABVS system also supports handheld ultrasound for innovative breast imaging applications, such as fatty tissue imaging, eSie Touch elasticity imaging and biopsy procedures.
Siemens will also be highlighting their proprietary eSieScan workflow protocols on the ACUSON S2000 system. Allowing the user to focus on patient care, not system interaction, eSieScan workflow protocols automatically guide the operator through the exam process by anticipating and executing exams based on customizable programs. Major modes, such as 2D, color, PW Doppler or M-mode can be automatically activated along with a variety of sub modes, such as eSie Touch elasticity or Cadence contrast pulse sequencing technology for contrast studies. (At this time, the FDA has cleared ultrasound contrast agents only for use in LVO.)
eSieScan protocols are available for vascular, breast and OB exams, but an unlimited number of user-defined protocols can be added to the system. By reducing the interaction of the user with the system, eSieScan protocols may lead to the reduction of repetitive stress injuries while at the same time increase the consistency and predictability of the exam content for the physician.
For more information: www.usa.siemens.com/healthcare


 April 09, 2026
April 09, 2026 









