September 20, 2018 — At the Transcatheter Cardiovascular Therapeutics (TCT) annual meeting, Sept. 21–25 in San Diego, Philips is showcasing its latest solutions in interventional cardiology. These solutions combine imaging systems, planning and navigation software, and specialized diagnostic and therapeutic devices, helping clinicians conduct procedures that are fast, efficient and personalized.
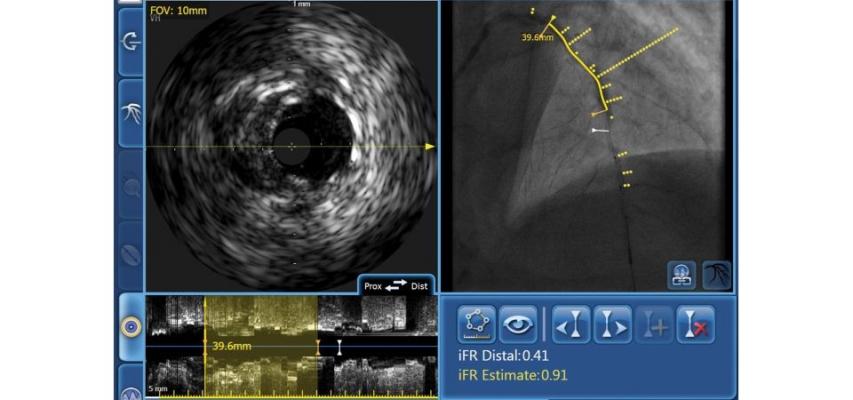
Philips will showcase its latest innovations in image-guided therapy, addressing key concerns of cardiologists such as complex percutaneous coronary interventions (complex PCI), which account for a significant proportion of PCI procedures that treat the narrowing of coronary arteries. Azurion, Philips’ next generation image-guided therapy platform, provides clinicians with the control needed to perform efficient procedures. CoreVision, which can be seamlessly combined with Azurion, is the only system, according to Phiips. that intelligently integrates X-ray angiography imaging, intravascular ultrasound (IVUS) and instant wave-free ratio (iFR) in a single view. Combining Philips’ imaging systems, planning and navigation, and smart devices, physicians can take advantage of an integrated solution that supports improved care for complex PCI patients.
This year at TCT, Philips will also showcase the recently introduced Epiq CVxi interventional cardiovascular ultrasound system. Specifically designed for use in the cath lab, the Epiq CVxi with EchoNavigator streamlines communication between the interventional cardiologist and the echocardiographer during complex interventional exams. Combining live ultrasound and X-ray information into one intuitive view, EchoNavigator helps interventional cardiologists oversee procedures along with the location of key anatomical structures. In addition, MultiVue provides more flexibility when using 3-D during diagnostic or interventional procedures as the clinician can see multiple views at once.
Also on show will be the compact and portable Philips CX50 ultrasound system, which has been upgraded with new software features and compatibility with additional transducers, including the latest X8-2t TEE transducer for transesophageal echocardiography. TEE places the ultrasound transducer close to the heart to enable confident diagnosis and treatment of structural heart diseases. Now, customers will be able to use a wider range of transducers, including the X8-2t TEE, across both their mobile and cart-based ultrasound systems.
For more information: www.usa.philips.com/healthcare


 June 02, 2026
June 02, 2026 









