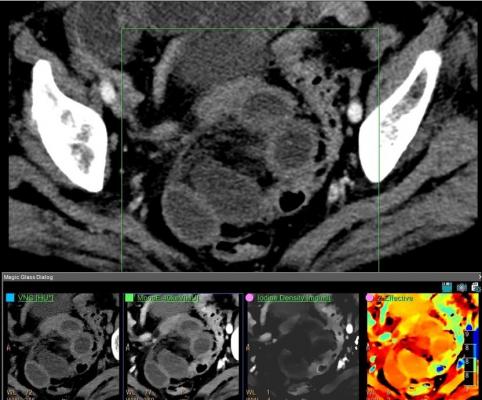
December 11, 2017 — At the 2017 Radiological Society of North America (RSNA) Annual Meeting, Philips unveiled the IQon Elite Spectral CT, designed to provide clinicians with increased diagnostic certainty in every scan. The system, a new configuration of the company's first spectral detector-based computed tomography (CT) scanner, debuts a suite of features that will enhance the clinical benefits of the IQon platform to improve diagnostic confidence while enhancing the patient experience, and supporting the needs of emergency/trauma and oncology care.
This new configuration provides a number of enhanced features and benefits, including:
- Enhanced diagnostic certainty - The IQon Elite’s faster reconstruction speeds and better visualization of bone marrow pathology enhance diagnostic confidence within emergency and trauma departments, providing the opportunity to reduce the need for subsequent scans. Its ability to estimate electron density provides enhanced tissue characterization, enabling a new level of diagnostic certainty for oncology clinicians and their patients.
- Increased patient volume - IQon Elite has been shown to double patient throughput to 200 patients per day (based on a 16-hour clinical shift). This is an important benchmark for healthcare facilities experiencing a high volume of patients, especially in the emergency department where CT can be a commonly used imaging modality.
- Enhanced clinical breadth – Its new radiation therapy planning couch and bariatric table top allow larger patients to be scanned with increased positioning controls.
The IQon Elite Spectral CT scanner will be available globally during the first quarter of 2018.
For more information: www.usa.philips.com/healthcare



 May 19, 2026
May 19, 2026 









