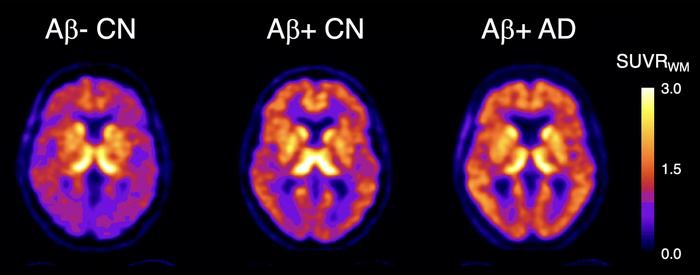
18F-SMBT-1 PET studies showed that beta-amyloid+ Alzheimer’s disease (AD) patients, but most importantly, beta-amyloid+ controls (CN) have significantly higher regional 18F-SMBT-1 binding than beta-amyloid- CN, with 18F-SMBT-1 retention highly associated with beta-amyloid burden. These findings suggest that increased 18F-SMBT-1 binding is detectable at the preclinical stages of beta-amyloid accumulation. Image created by Victor Villemagne, MD, Professor of Psychiatry at the University of Pittsburgh.
October 20, 2022 — A new highly selective PET imaging agent can detect the presence of overexpressed monoamine oxidase-B (MAO-B) in cognitively unimpaired individuals with high beta amyloid (Aβ)—one of the earliest signs of Alzheimer’s disease—according to research published in the October issue of The Journal of Nuclear Medicine. The radiotracer, 18F-SMBT-1, allows for a better understanding of the role of inflammation in Alzheimer’s disease, which can enable more accurate staging and prognosis at earlier stages.
Brain inflammation that accompanies Alzheimer’s disease involves reactive astrocytes, which are cells that overexpress MAO-B. The newly developed 18F-SMBT-1 radiotracer is highly selective for MAO-B and as a result has increased binding to reactive astrocytes. “This increased binding suggests that 18F-SMBT-1 can potentially be used as a surrogate marker to detect reactive astrogliosis in Alzheimer’s disease,” noted Victor Villemagne, MD, professor of psychiatry at the University of Pittsburgh in Pittsburgh, Pennsylvania.
The study aimed to characterize 18F-SMBT-1 binding to reactive astrocytes across the Alzheimer’s disease continuum. Study participants included three clinical groups: 57 cognitively unimpaired controls, 12 subjects meeting criteria for mild cognitive impairment (MCI), and eight subjects meeting criteria for Alzheimer’s disease.
Participants underwent several types of imaging, including 18F-SMBT-1 PET, Aβ PET, tau PET, and MRI. Images were normalized and statistical analyses conducted to assess 18F-SMBT-1 binding in relation to Aβ and tau pathology burden. 18F-SMBT-1 was found to be highly correlated with Aβ burden, and much less with tau burden.
The three clinical groups were then classified based on their Aβ status (either as Aβ+ or Aβ-). No significant differences in 18F-SMBT-1 binding were found among Aβ- participants in the control and MCI groups. In the Aβ+ subjects with Alzheimer’s disease, 18F-SMBT-1 binding was significantly higher. Most importantly, 18F-SMBT-1 binding was significantly higher in the Aβ+ control group as compared to Aβ- control group.
“It’s of note that the brain regions where we saw this higher 18F-SMBT-1 binding in the control group are regions known for early Aβ deposition. This suggests that reactive astrocytes are associated with early Aβ deposition at the preclinical stages of Alzheimer’s disease and likely play a role over clinical progression,” said Villemagne.
He continued, “Implementation of 18F-SMBT-1 will clarify the role of reactive astrogliosis in neurodegenerative conditions, not just Alzheimer’s disease and its potential independent and/or synergistic effects on pathology, neurodegeneration, cognition, and disease progression. This has the potential to define and refine the diagnostic, staging and prognostic roles of reactive astrogliosis in these conditions.”
For more information: www.snmmi.org
Related Alzheimer's Content:
FDA Grants Accelerated Approval for Alzheimer’s Drug Aduhelm
VIDEO: Researchers Use MRI to Predict Alzheimer's Disease
Brain Iron Accumulation Linked to Cognitive Decline in Alzheimer's Patients
Good Results for Alzheimer’s Imaging Agent
NIH Augments Large Scale Study of Alzheimer’s Disease Biomarkers
Alzheimer’s Association Launches New Website for IDEAS Study
PET Tracer Gauges Effectiveness of Promising Alzheimer's Treatment


 May 27, 2026
May 27, 2026 









