
Greg Freiherr has reported on developments in radiology since 1983. He runs the consulting service, The Freiherr Group.
Digital Technology Pushes PET In New and Old Directions
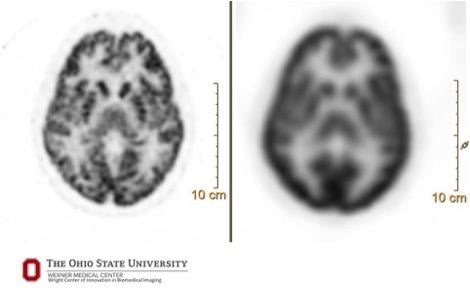
A 90-second brain acquisition with FDG radiotracer — comparison of digital (Vereos, left, 1 mm) and conventional (Gemini TF, 4 mm) images.
Digital technology is opening remarkable opportunities for clinical positron emission tomography (PET) about which research is only beginning to hint.
“Twenty years ago we were excited that we could see a lesion. Now we want to understand its underlying biologic heterogeneity,” said Michael Knopp, M.D., a radiology professor at Ohio State University whose research with Philips’ digital PET/CT, called Vereos, is exploring applications within and beyond traditional clinical areas.
The digital technology underlying Vereos can provide the details that may escape analog systems, said Piotr Maniawski, director of clinical science for nuclear medicine at Philips Healthcare. Visualization using 4 mm cubes, which are typically delivered by analog systems, makes small lesions look spherical, he said. The very small voxels in digital images better visualize shapes and texture.
“Seeing if the texture is changing helps characterize the lesion,” said the Philips clinical director. The metabolism inside the lesion, he said, may be much different, depending on the location.
In a paper published May 2017 in the journal Contrast Media and Molecular Imaging, Knopp, who is the Novartis chair of Imaging Research at Ohio State University, and colleagues at Ohio State summarized how digital PET enables advanced functional tumor imaging.
A Digital Twist
Much like radiography began, PET started as an analog modality. Instead of film, PET relied on photomultiplier tubes (PMTs). In analog systems, light generated by a single scintillating crystal is channeled to multiple PMTs. In Vereos, light generated by a single scintillating crystal is channeled to its own detector.
Vereos’ digital detection is built on digital photon counting (DPC) technology, whereby crystals and sensors are coupled “one-to-one,” said Maniawski, who has worked with Knopp and others to assess the clinical capabilities conferred by Vereos’ digital technology. Light flashes produced by specific crystals are channeled directly to individual digital sensors. In stark contrast to analog technology, which accumulates signals from light flashes in analog detectors until reaching trigger points, “there is no light sharing,” Maniawski said. “The moment the detector registers this light, we know precisely which crystal produced it.”
Reconstruction algorithms onboard Vereos mathematically reconstruct digital PET into detailed images. These appear “more rich and precise” than those made using analog-based PET systems, Knopp said, due to the increased density of the data. The OSU researcher likened the benefit of data density to the improvement of smartphone images as those pictures gain more data density.
“When you pull up Google maps, the picture might look fuzzy at first and then as the data come in, it will look sharp and brilliant,” Knopp said. “This happens because the data density changes.”
Maniawski explained that voxels in Vereos images are densified with data collected by the 23,000 solid-state sensors from individual scintillation events. This added data density allows Vereos to package data into voxel volumes of 2 mm — or even 1 mm — cubes. This data density gives shape and texture to structures in the images.
Bringing Together Old and New
The game-changing appearance of Vereos’ images can be challenging, Knopp noted.
Because digital images show more detail, they may show lesions and features that might not be seen with analog technology. When comparing current digital images to past analog ones, the question arises: Were lesions now visible not seen previously because the technology could not see them? Or have they just recently occurred?
The answers to these questions can directly impact the management of patients being monitored for disease recurrence.
To deal with this incompatibility, Philips offers a feature in Vereos that reconstructs digital data as if they were acquired on an analog system. This is done with reconstruction algorithms, Maniawski said. These algorithms harmonize the digital data.
Vereos users who choose this option “end up with two images from the same data — a conventional looking image and a digital one,” Maniawski said.
Vereos can create a still picture and visualize changes over time. In the mid- to late-1980s, early developers of PET often acquired data dynamically. Serial acquisitions came into vogue when PET/CT imaging entered the mainstream. But dynamic imaging, similar to short video clips, can provide clinical information not found in static images.
Editor’s Note: This is the fifth in a series of blogs on the advances of digital PET/CT. The first is entitled How Digital PET/CT Can Improve Clinical Care; the second Why – And How – Digital PET Is Better Than Analog; the third Digital PET Balances Scan Time and Resolution; the fourth What Precision Means To PET.
Related Content:


 May 08, 2026
May 08, 2026 









