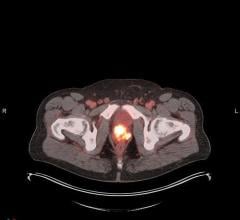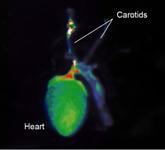
November 13, 2009 – A new imaging agent for measuring and monitoring cathepsin B activity associated with disease progression and therapeutic response in vivo will be released at the American Heart Association meeting in Orlando this weekend.
VisEn Medical Inc. said it is launching its new Cat B 750 FAST agent. Cathepsin B expression is well known as a key biomarker and therapeutic target in a range of diseases, including atherosclerosis, oncology, and arthritis. The new agent is expected to have a significant impact in cardiovascular disease research.
Cathepsin B proteases play an important role in disease progression and host response in several disease areas, and are key biomarkers in assessing and characterizing disease states. The new Cat B 750 FAST agent is designed to complement in vitro and in vivo protease readouts from cell-based assays and VisEn’s existing in vivo agent product lines, providing earlier imaging time points and narrower specificity profiles of cathepsin B within the cathepsin family of proteases.
The new Cat B 750 FAST agent represents an additional protease-sensing agent developed from VisEn’s new FAST (Fluorescent Activatable Sensor Technology) in vivo agent platform. FAST agents are based on high-performance activatable smaller molecules that combine advanced biomarker specificity with optimized biocompatibility and rapid pharmacokinetics. These benefits allow for precise measurement and monitoring of key disease biomarkers in vivo.
VisEn also announced the commercial availability of its ReninSense680 FAST agent for localized in vivo measurement of changes in renin-angiotensin system (RAS) involved in hypertension and some neurological diseases.
All VisEn agents and labels are designed for in vivo biomarker quantification using VisEn’s Fluorescence Molecular Tomography (FMT) imaging systems, or for general use along with other non-quantitative fluorescence in vivo imaging systems. VisEn agents are also designed to enable complementary in vitro biomarker readouts in cells and tissues using standard fluorescence microscopy and cellular-based imaging systems. With a focus on translational research and results, all of VisEn’s technologies are designed to generate translational data linking pre-clinical research into clinical medicine.
For more information: www.visenmedical.com


 May 29, 2026
May 29, 2026