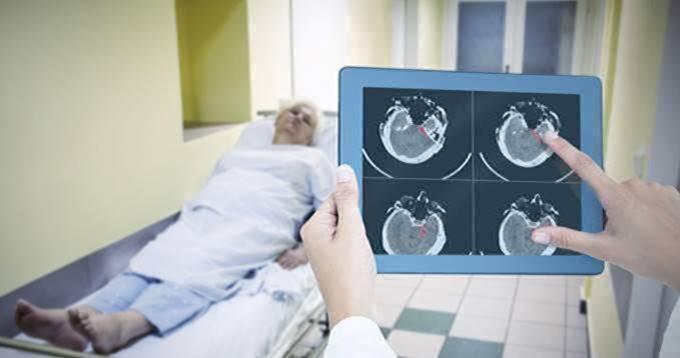
November 7, 2018 – MaxQ AI announced that its Accipio Ix intracranial hemorrhage (ICH) detection software has received 510(k) clearance from the U.S. Food and Drug Administration (FDA). The clearance paves the way for healthcare providers and physicians in acute care settings to have access to this artificial intelligence (AI) software designed to aid in prioritizing the clinical assessment of adult non-contrast head computed tomography (CT) cases that exhibit indications of intracranial hemorrhage (ICH), commonly known as a brain bleed.
Earlier this year, MaxQ announced CE Mark approval and commercial availability in the European Union. The software leverages artificial intelligence technology to automatically analyze non-contrast head CT images without workflow impact to the reader, altering the original series or storing protected health information (PHI). The AI-powered Accipio Ix, part of MaxQ’s Accipio Insight platform, is designed to be highly sensitive to the presence of ICH, identifying and prioritizing patients with ICH for the treating physician. It provides a capability for rapid escalation and prioritization of the patient, and can be natively integrated into CT and picture archiving and communication systems (PACS) using the imaging industry-standard DICOM, installed both on-premise and cloud-capable.
MaxQ will be demonstrating Accipio Ix – along with the full suite of Accipio platform solutions – during the Radiological Society of North America (RSNA) 2018 Annual Meeting, Nov. 25-30 in Chicago.
For more information: www.maxq.ai


 May 05, 2026
May 05, 2026 









