December 24, 2019 — LG Electronics Business Solutions introduced a larger, higher-resolution radiology and surgical monitors designed to offer hospitals and clinics improved work efficiency, advanced performance, expanded screen real estate and clean, minimalist designs. Demonstrated at RSNA 2019, the three new LG medical displays further expand the company’s diverse healthcare portfolio.
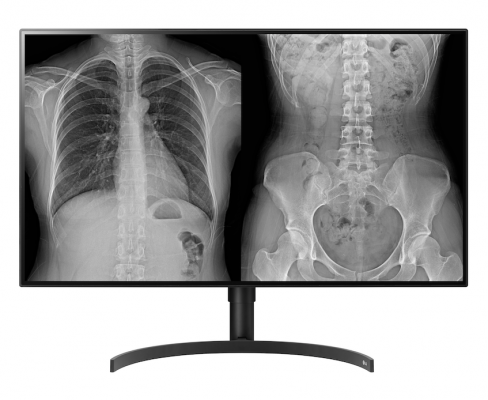
LG 31.5-inch 8MP Radiology Monitor
LG’s new 8-megapixel radiology monitor (model 32HL512D – FDA 510k Class II approval is pending) has a larger screen than its 27-inch predecessor and employs LG NanoCell IPS display technology for image quality optimized for accurate reviews. While reducing unwanted light wavelengths, the NanoCell IPS display enhances color purity to reproduce precise images that can be clearly seen from almost any vantage point. This 31.5-inch 8MP monitor is lightweight and cost-effective, while being designed to deliver excellent image quality. It features out-of-the-box DICOM calibrated to ACR- AAPM-SIIM guidelines. With two DisplayPort video inputs, this monitor can be operated as two 4MP screens or as a single 8MP screen.
LG 31.5-inch 4K Surgical Monitor
LG’s new 4K surgical monitor (model 32HL710S) features a large ultra-high-definition display that leverages LG’s innovative technologies to provide highly detailed images,
greater ease of use, and reliability necessary for the O.R. This monitor is engineered to provide low latency from video source to display. Its backlight brightness monitoring and control produce consistent flicker-free video. Model 32HL710S can be used with a wide variety of endoscopy cameras to provide remarkably smooth and accurate up-scaling of Full-HD video.
The monitor also supports high dynamic range (HDR 10) content, delivering impressive contrast and colors when connected to an endoscope camera or other compatible devices. The advanced HDR 10 video format gives medical professionals a clear picture that’s easy to see and interpret. LG’s surgical monitor has 4PBP and PIP modes as well as multiple video input ports to display visual information from several devices simultaneously. This enhances work efficiency by making it possible to view a comprehensive range of data on a single screen.
For improved flexibility, the 32HL710S provides the Mirror and Rotate functions. It also features easily- cleanable protective glass and has lightweight sleek design with a magnetic cover for precision cable management.
LG 24-inch Health Care All-in-One Thin Client
LG’s first cloud device for the health care market, model 24CK560N is the company’s user-friendly solution designed to improve workflow for busy hospital environments. A complete product in its own right, the 24CK560N features a DisplayPort video output for connection of second monitor. Its IPS panel produces vibrant images with consistent colors and contrast from any viewing angle, meaning groups can gather around and clearly see any content on screen.
LG’s Thin Client 24CK560N can be quickly repositioned according to the user’s needs with swivel-, pivot- and height-adjustability. What’s more, the all-in-one system represents a space-efficient replacement for existing PC and monitor setups, freeing up desk space and helping to create a less cluttered environment.
The product also been carefully designed to support the high level of hygiene required in medical facilities. Its white cabinet makes it easy for users or contracted cleaners to see and remove any dust or other foreign substances. Its fan-less design ensures a quiet performance that won’t distract users or patients. The cloud device can be attached to a medical cart for maximum portability, letting staff securely log in and access information and resources from anywhere in the hospital. Model 24CK560N complies with a number of medical standards (IEC6060, CE MDD and FDA Class I), assuring suitability for healthcare settings.
For more information: www.lg.com/us


 April 23, 2026
April 23, 2026