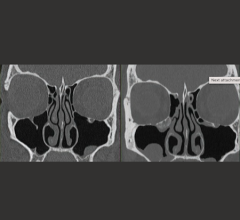
October 1, 2015 — Infinitt now offers an integrated workflow reporting and tracking solution--an optional add-on module to the Infinitt RIS-- that empowers providers to implement a successful lung screening program in accordance with best practice guidelines.
Infinitt CT Lung follows the recommendations of the American College of Radiology (ACR), providing the tools you need to attain accreditation as an ACR-Designated Lung Cancer Screening Center participant. In addition, the ACR offers customizable tools to help you promote your facility to patients and referring physicians.
Features:
• Provides Lung Reporting and Data System (Lung-RAD) conformant reports and letter management; reminder letters in MS Word are fully customizable.
• Designed to improve patient interaction and standardize communications; also as a quality assurance tool to standardize lung cancer screening, reporting and management.
• Biopsy Result entry enables lab results to be included with radiologist’s findings.
• Seamless integration with Infinitt DoseM for radiation dose data tracking and analysis.
• Simplifies submission of results to the American Lung Cancer Society Registry.
• Includes secondary result findings with appropriate tracking and letter management.
• Enables you to evaluate your Lung Screening Program relative to your own key performance indicators.
The Centers for Medicare and Medicaid Services (CMS) issued a national coverage decision (NCD) for low dose computed tomography (LDCT) lung cancer screening on Feb. 5, 2015. For information on lung cancer screening coverage and reimbursement, visit the Lung Cancer Screening Resources page.
For more information: www.infinittna.com


 May 01, 2026
May 01, 2026