July 3, 2018 — HeartFlow Inc. announced that UnitedHealthcare now covers the HeartFlow FFRct Analysis, extending access to their 45 million beneficiaries. With this new coverage, more than 235 million people in the United States now have access to the HeartFlow Analysis.
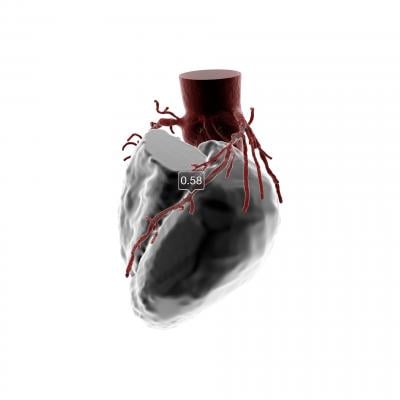
The non-invasive technology uses a coronary computed tomography angiogram (CTA) to create a personalized 3-D model of the heart and simulate blood flow in order to help clinicians diagnose and treat people with suspected coronary artery disease (CAD).
In a new coverage decision, UnitedHealthcare also chose to adopt the medical policy for coronary CTA from eviCore, an evidence-based specialty benefits management company. The policy allows for the use of a coronary CTA as a first-line test for symptomatic individuals, removing the need for a standard stress test prior to conducting a coronary CTA. In addition, the HeartFlow Analysis is now approved to further assess coronary disease seen on a coronary CTA that is of uncertain physiological significance.
CAD affects 16.8 million Americans and develops when the coronary arteries narrow, reducing blood flow to the heart and potentially causing chest pain (angina), heart attack (myocardial infarction) and death.
Clinicians diagnosing someone with suspected CAD want to know as definitively as possible if the individual has a significant blockage in their coronary arteries. They also want to know the impact of that blockage on blood flow so they can best determine which treatment pathway is appropriate (e.g., medical management, stenting or coronary artery bypass grafting).
Data from a patient’s non-invasive coronary CTA are securely uploaded from the hospital’s system to the cloud. HeartFlow leverages deep learning to create a personalized, digital 3-D model of the patient’s coronary arteries. The HeartFlow Analysis then uses powerful computer algorithms to solve millions of complex equations to simulate blood flow and assess the impact of blockages on coronary blood flow. The service is provided via a secure online interface to offer actionable information to enable clinicians to determine the optimal course of treatment.
For more information: www.heartflow.com


 May 19, 2026
May 19, 2026 









