November 17, 2014 — The U.S. Food and Drug Administration (FDA) has cleared Siemens' syngo.CT Single Source Dual Energy software package, that enables spectral imaging using which is designed to operate with CT images acquired with Siemens single source CT scanners.
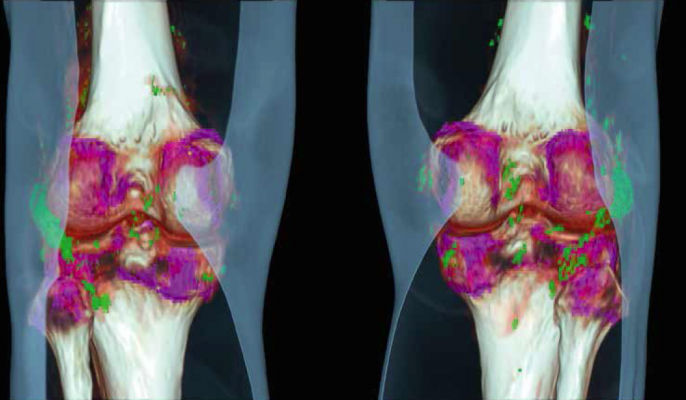
The various materials of an anatomical region of interest have different attenuation coefficients, which depend on the used energy. These differences provide information on the chemical composition of the scanned body materials. syngo.CT Single Source Dual Energy combines images acquired with low and high energy spectra to visualize this information.
Depending on the organ of interest, the user can select and modify different application classes or parameters and algorithms. syngo.CT Single Source Dual Energy Software Package is a post processing application package consisting of several post processing application classes that can be used to improve visualization of various materials in the human body.
Multiplanar reformations (MPR) of the volume are shown in three image segments, which are initialized as sagittal, coronal and axial view. After arriving at an initial diagnosis on the basis of the CT-images, the user can choose one of the following application classes: monoenergetic imaging, gout evaluation, brain hemorrhage, liver VNC, monoenergetic plus, bone marrow and kidney stones. Kidney stones imaging is possible with the visualization of the chemical composition of the kidney stones and especially the differentiation between uric acid and non-uric acid stones.
After loading the two reconstructed image datasets acquired with two different X-ray spectra into the software, a registration is performed in case the image data sets are not acquired simultaneously, to compensate for potential motion effects. They are then displayed using linear blending with selectable mixing ratio and color scale.
The software operates on the most recent version syngo.via client server
For more information: www.healthcare.siemens.com


 May 19, 2026
May 19, 2026 









