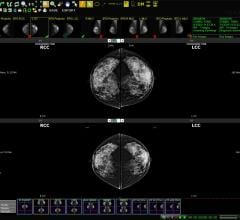
December 4, 2009 - Breast-Specific Gamma Imaging (BSGI) is a molecular breast imaging technique increasingly being used in the diagnostic workup of patients with indeterminate mammograms and has demonstrated the ability to detect cancers missed by mammography and ultrasound. However, broad acceptance of this imaging technique has been limited due to the inability to perform minimally invasive, image-guided biopsy procedures under this modality.
One technique that may break through this limitation is stereotactic BSGI needle guidance, according to findings presented by Cynthia Lorino, M.D., of the Montgomery Breast Center of Montgomery, Ala. at the annual meeting of the Radiological Society of North America (RSNA).
In this study, the GammaL c device, an accessory to the Dilon 6800 Gamma Camera, provided accurate lesion localization and facilitated needle-guided biopsy. The stereotactic lesion localization system was developed to allow BSGI-guided, vacuum-assisted needle biopsy. The purpose of the study was to examine the clinical viability of the system.
Patients who were routinely scheduled for a needle biopsy based on clinical findings and also had a focal abnormality on BSGI were invited to participate in the clinical trial of the GammaL c system. In the study, 17 patients of variable breast sizes patients with lesions ranging from 4 mm to 70 mm underwent BSGI-guided vacuum-assisted needle biopsies. All of the biopsy results were concordant with known Sestamibi avid pathologies successfully demonstrating the clinical accuracy of this guidance system. In addition, in some cases, both the BSGI diagnostic examination and Gamma-Guided biopsy were conducted in a single day using only one administration of Sestamibi.
The authors wrote in their conclusion, "The GammaLoc stereotactic biopsy apparatus provides an accurate, convenient means for conducting vacuum-assisted needle biopsy of breast abnormalities."
The GammaL c accessory is pending FDA clearance.
For more information: www.dilon.com


 July 07, 2021
July 07, 2021