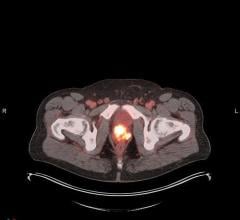
October 8, 2009 - Bayer Schering Pharma AG, Germany, will provide its development candidate florbetaben, a PET (positron emission tomography) tracer, to the Swiss-based biopharmaceutical company AC Immune SA, to support a clinical trial in the field of Alzheimer´s disease (AD). This study is conducted to develop a therapy option for the treatment of AD. Bayer´s novel PET tracer florbetaben will be applied for imaging of beta-Amyloid deposition in the brain of patients undergoing the phase I clinical testing of AC Immune´s Alzheimer´s vaccine ACI-24. In this collaboration, florbetaben will be tested for the very first time in the development of a potential therapeutic for Alzheimer’s patients.
"Bayer Schering Pharma has already demonstrated the potential of florbetaben to image beta-Amyloid deposition in the brain in its own phase II study," said Dr. Thomas Balzer, Head of Global Clinical Development Therapeutic Area Diagnostic Imaging at Bayer Schering Pharma. "The cooperation with AC Immune enables the collection of valuable clinical data for florbetaben in patients treated with AC Immune’s novel therapeutic vaccine."
"This collaboration with Bayer Schering Pharma adds further value to our Alzheimer´s vaccine ACI-24," said Prof. Andrea Pfeifer, CEO of AC Immune. "The adoption of a diagnostic imaging substance visualizing the deposition of beta-Amyloid that is targeted by our vaccine can be an important parameter for dose selection, and will provide useful complementary data. There is a key trend in the industry to co-develop therapy guiding diagnostics alongside drugs for patients. We look forward to working with Bayer´s experts in this ground-breaking collaboration."
Currently, there is no diagnostic test on the market that can detect beta-Amyloid deposition in the brain - a pathological hallmark of Alzheimer´s disease - in patient´s lifetime. Today, the clinical diagnosis of AD is based on cognitive tests, Magnetic Resonance Imaging (MRI) and Computerized Tomography (CT) scans to exclude other dementia diseases. Unfortunately, clinical diagnosis is often made too late and does not always correlate with post-mortem diagnosis. A good diagnostic assay should also help to better evaluate the effect of new treatments in clinical trials as well as correlate better with existing pathological and memory markers. A new diagnostic tool to detect beta-Amyloid in the brain in vivo might also be beneficial in detecting the disease earlier, before the symptoms are too advanced so treatment could be started earlier.
Florbetaben (18 F) is an inlicensed 18F-labeled PET tracer that specifically binds to deposition of beta-Amyloid. These depositions (plaques) consist of proteins that accumulate in the brain and are a pathological hallmark of Alzheimer’s disease. As the aggregation of the beta-Amyloid protein in the brain is also a key target for new therapeutic treatments under development, florbetaben might also be able to support the development of these new treatment approaches. A phase II study showed that patients with clinical diagnosis of Alzheimer´s disease could be differentiated from age-matched healthy volunteers on the basis of florbetaben uptake pattern in the brain. The results of this study were presented at the International Conference on Alzheimer’s diseases (ICAD) in Vienna, Austria in July 2009.
Find more information: www.viva.vita.bayerhealthcare.com


 May 29, 2026
May 29, 2026