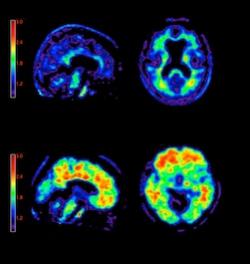
April 6, 2012 – The U.S. Food and Drug Administration (FDA) has approved the first diagnostic agent to image Alzheimer’s disease beta-amyloid neuritic plaques in the living brain. Previously, the only way to confirm diagnosis of these plaques was from post-mortem biopsies.
Eli Lilly and Company and Avid Radiopharmaceuticals Inc., a wholly owned subsidiary of Lilly, said the FDA cleared the commercial release of florbetapir (Amyvid), a positron emission tomography (PET) radioactive tracer agent indicated for brain imaging of beta-amyloid plaques in patients with cognitive impairment who are being evaluated for Alzheimer's disease and other causes of cognitive decline.[1] Amyvid binds to amyloid plaques, a hallmark characteristic of Alzheimer's disease,[2, 3, 4] and is detected using PET scan images of the brain.[1]
A negative Amyvid scan indicates sparse to no amyloid plaques are currently present, which is inconsistent with a neuropathological diagnosis of Alzheimer's disease and reduces the likelihood that a patient's cognitive impairment is due to Alzheimer's disease.[2, 5] A positive Amyvid scan indicates moderate to frequent amyloid plaques are present; this amount of amyloid plaque is present in patients with Alzheimer's disease, but may also be present in patients with other types of neurologic conditions and in older people with normal cognition.[1, 4, 6]
It is important to note that Amyvid is an adjunct to other diagnostic evaluations. A positive Amyvid scan does not establish a diagnosis of Alzheimer's disease or other cognitive disorder. Additionally, the safety and effectiveness of Amyvid have not been established for predicting development of dementia or other neurologic conditions or monitoring responses to therapies.[1]
Alzheimer's disease is one of several possible causes of cognitive decline. Alzheimer's disease and other causes of cognitive impairment share many overlapping symptoms, including memory, visuospatial ability, executive function, behavior and language deficiencies.[4, 7] Because a definitive diagnosis of Alzheimer's disease is usually determined upon autopsy,[8] physicians rely on medical history, clinical examination and a variety of diagnostic tools when evaluating patients.[4]
"It's estimated that one in five patients clinically diagnosed with probable Alzheimer's disease during life do not end up having Alzheimer's disease pathology upon autopsy,"[9, 10] said Daniel Skovronsky, M.D., Ph.D., president and CEO of Avid, and global brand development leader for Amyvid at Lilly. "The approval of Amyvid offers physicians a tool that, in conjunction with other diagnostic evaluations, can provide information to help physicians evaluate their patients."
Amyvid was evaluated in three clinical studies that examined images from healthy adult patients as well as patients with a range of cognitive disorders, including some terminally ill patients who had agreed to participate in a post-mortem brain donation program. Based on the results of study one, measurements of post-mortem cortical amyloid burden correlated with median Amyvid scores (r=0.78; P<0.0001). In the second study, using the majority interpretation of five readers, Amyvid PET showed 96 percent sensitivity and 100 percent specificity in patients who received an Amyvid PET scan within one year of death. Across all readers and all autopsied patients, Amyvid PET demonstrated median sensitivity of 92 percent (range 69 to 95 percent) and specificity of 95 percent (range 90 to 100 percent) for readers trained in person (study two), and median sensitivity of 82 percent (range 69 to 92 percent) and specificity of 95 percent (range 90 to 95 percent) for readers trained using an electronic media-based training (study three). Additionally, inter-reader reproducibility analysis for all images in study three showed an overall Fleiss' kappa statistic of 0.83 (95 percent CI: 0.78 to 0.88).
The most common adverse reactions reported in clinical trials were headache (1.8 percent), musculoskeletal pain (0.8 percent), fatigue (0.6 percent), nausea (0.6 percent), anxiety (0.4 percent), back pain (0.4 percent), increased blood pressure (0.4 percent), claustrophobia (0.4 percent), feeling cold (0.4 percent), insomnia (0.4 percent) and neck pain (0.4 percent).[1]
"Florbetapir gives patients with cognitive decline, their families and the physicians who treat them, more information about the amyloid plaques that may be found in their brain," said Edward Coleman, M.D., professor of radiology, Duke University Medical Center. "This approval marks a great advancement in nuclear medicine practice, as it enables us to evaluate the presence or absence of moderate to frequent levels of amyloid plaques in a patient's brain. In conjunction with other tests, florbetapir may help give physicians additional information when evaluating patients for the cause of their cognitive decline."
Because Amyvid loses more than half of its radioactivity every two hours, Amyvid must be distributed directly from a radiopharmacy to the imaging centers where it will be administered within several hours. Beginning in June, a limited number of radiopharmacies will be distributing Amyvid with the goal of making the product available in more areas as soon as possible.
"The approval of Amyvid exemplifies Lilly's commitment to discovering and developing innovative products for many of the world's unmet medical needs," said Alex Azar, president, Lilly USA. "We are working hard with our manufacturing partners to increase production of Amyvid and will notify the community as it becomes available in more markets."
Amyvid images should be interpreted only by readers who have successfully completed Amyvid reader training. Lilly has worked collaboratively with the FDA and nuclear medicine experts to identify the appropriate ways to support accurate and consistent interpretation of Amyvid scans by imaging physicians. These efforts resulted in the development and validation by Lilly of both an online and in-person reader-training program for physicians using Amyvid. Errors may occur in the estimation of plaque density during image interpretation.
About Amyvid
Amyvid is a radioactive diagnostic agent tagged with a radioisotope called fluorine-18 (F-18). Once Amyvid is injected into a vein, it travels through the bloodstream and into the brain, binding to amyloid plaques. Amyvid produces a positron signal, which is detected by a PET scanner and used to create a brain image. A radiologist, who should have successfully completed Amyvid reader training, then interprets the image to evaluate for the presence or absence of significant amyloid plaques (i.e., moderate to frequent levels of neuritic plaques) in the brain. This information is reported back to the referring physician, who then determines the next steps in the evaluation and management of the patient.
Amyvid is supplied in 10 mL, 30 mL or 50 mL multidose vials containing 500-1,900 MBq/mL florbetapir F-18.
Indication
Amyvid is a radioactive diagnostic agent for PET imaging of the brain to estimate beta-amyloid neuritic plaque density in adult patients with cognitive impairment who are being evaluated for Alzheimer's disease and other causes of cognitive decline. A negative Amyvid scan indicates sparse to no neuritic plaques and is inconsistent with a neuropathological diagnosis of Alzheimer's disease at the time of image acquisition; a negative scan result reduces the likelihood that a patient's cognitive impairment is due to Alzheimer's disease. A positive Amyvid scan indicates moderate to frequent amyloid neuritic plaques; neuropathological examination has shown this amount of amyloid neuritic plaque is present in patients with Alzheimer's disease, but may also be present in patients with other types of neurologic conditions as well as older people with normal cognition. Amyvid is an adjunct to other diagnostic evaluations.
Limitations of Use
A positive Amyvid scan does not establish a diagnosis of Alzheimer's disease or other cognitive disorder. Additionally, the safety and effectiveness of Amyvid have not been established for predicting development of dementia or other neurologic condition or monitoring responses to therapies.[1]
Risk for Image Misinterpretation and other Errors
Errors may occur in the Amyvid estimation of brain neuritic plaque density during image interpretation.
Image interpretation should be performed independently of the patient's clinical information. The use of clinical information in the interpretation of Amyvid images has not been evaluated and may lead to errors. Other errors may be due to extensive brain atrophy that limits the ability to distinguish gray and white matter on the Amyvid scan as well as motion artifacts that distort the image.
Amyvid scan results are indicative of the brain neuritic amyloid plaque content only at the time of image acquisition and a negative scan result does not preclude the development of brain amyloid in the future.
For more information: www.lilly.com
References:
1. Amyvid Prescribing Information. April 6, 2012.
2. Hyman BT, Phelps CH, Beach TG, et al. “National Institute on Aging–Alzheimer's Association guidelines for the neuropathologic assessment of Alzheimer's Disease.” Alzheimers Dement. 2012;8:1-13.
3. Mirra SS, Heyman A, McKeel D, et al; and participating CERAD neuropathologists. “The Consortium to Establish a Registry for Alzheimer's Disease (CERAD): part II: standardization of the neuropathologic assessment of Alzheimer's disease.” Neurology. 1991;41(4):479–486.
4. Thies W, Bleiler L; Alzheimer's Association. “Alzheimer's Association report: 2012 Alzheimer's disease facts and figures.” Alzheimers Dement. 2012;8:131-168.
5. McKhann GM, Knopman DS, Chertkow H, et al. “The diagnosis of dementia due to Alzheimer's Disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's Disease.” Alzheimers Dement. 2011;7:263-269.
6. Sperling RA, Aisen PS, Beckett LA, et al. “Toward defining the preclinical stages of Alzheimer's Disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's Disease.” Alzheimers Dement. 2011;7(3):280–292.
7. Balasa M, Gelpi E, Antonell A, et al. “The Neurological Tissue Bank/University of Barcelona/Hospital Clínic NTB/UB/HC Collaborative Group. Clinical features and APOE genotype of pathologically proven early-onset Alzheimer Disease.” Neurology.2011;76(20):1720–1725.
8. Piccini A. CSF biomarkers. Open Nucl Med J. 2010;2:25-30.
9. Lim A, Tsuang D, Kukull W, et al. “Clinico-neuropathological correlation of Alzheimer's disease in a community-based case series.” J Am Geriatr Soc. 1999;47(5):564–569.
10. Petrovitch H, White LR, Ross GW, et al. “Accuracy of clinical criteria for AD in the Honolulu-Asia Aging Study, a population-based study.” Neurology. 2001;57(2):226–234.


 May 08, 2026
May 08, 2026 









