An estimated 24% of women in the United States will suffer from a pelvic floor disorder in her lifetime, with that varying significantly with age, as by the time women are 80 years old, up to 50% are affected. This number is expected to increase by over 70% by the year 2050, primarily due to an aging population.1 This makes them one of the most common health issues for women, and yet they are vastly underdiagnosed and undertreated.
Pelvic Floor Disorders
Pelvic floor disorders arise from injury to and/or dysfunction of the muscles or connective tissues that result in a variety of conditions, with the most common being pelvic organ prolapse and urinary and fecal incontinence. Women with significant pelvic floor dysfunction have a lower quality of life, and also are more likely to have their sexual function impacted. Not included under this umbrella term are other issues of the pelvic floor related to non-relaxation, so in reality the numbers given previously are even higher. Underlying causes include genetic predispositions and inherited diseases of connective tissue, vaginal birth, surgical procedures, sexual trauma, obesity, chronic constipation and other lifestyle factors.2
As with any disorder, raising awareness for healthcare professionals as well as the public is the first step. Once a pelvic floor disorder is suspected, proper diagnosis is critical in the evaluation and treatment process. Ultrasound has been in use for decades as a common diagnostic tool in women’s health, but adoption of newer advancements has been lagging behind scientific knowledge.3Magnetic resonance imaging (MRI) was the first tool to evaluate the anatomy of the pelvic floor, and in the early 1990s its use as a diagnostic tool added a significant amount of knowledge. But its inclusion in diagnostic protocols was limited by several factors including static supine images, radiation, cost and availability. By the early 2000s, 3-D ultrasound demonstrated clear advantages in imaging the pelvic floor and its dynamic function, and was subsequently shown to alter surgical approach to pelvic floor disorders in over 60% of cases in one study.4 But despite current widespread availability of 3-D ultrasound, this modality is still vastly under-recognized and under-utilized in the diagnostic evaluation.
In this article we will discuss the role of digital imaging of the pelvic floor for diagnosis and management of pelvic floor disorders including urinary incontinence, pelvic organ prolapse and obstetric injuries in the postpartum period.
Pelvic Organ Prolapse
Pelvic organ prolapse occurs when the pelvic floor no longer supports the normal positioning of the pelvic organs. This can lead to descent of the vaginal walls and/or uterus, which may range from mild and asymptomatic to more severe cases where quality of life is significantly impacted. While it can occur at any age, the peak incidence is for women in their eighth decade of life.
It is interesting that while 40-50% of women will have evidence of pelvic organ prolapse on physical exam, less than 5% self-report symptoms of vaginal bulging.5 But with pelvic floor relaxation, a significant contributing factor for the 1 in 4 women who develop urinary incontinence, the number of women affected by pelvic floor relaxation is even greater.
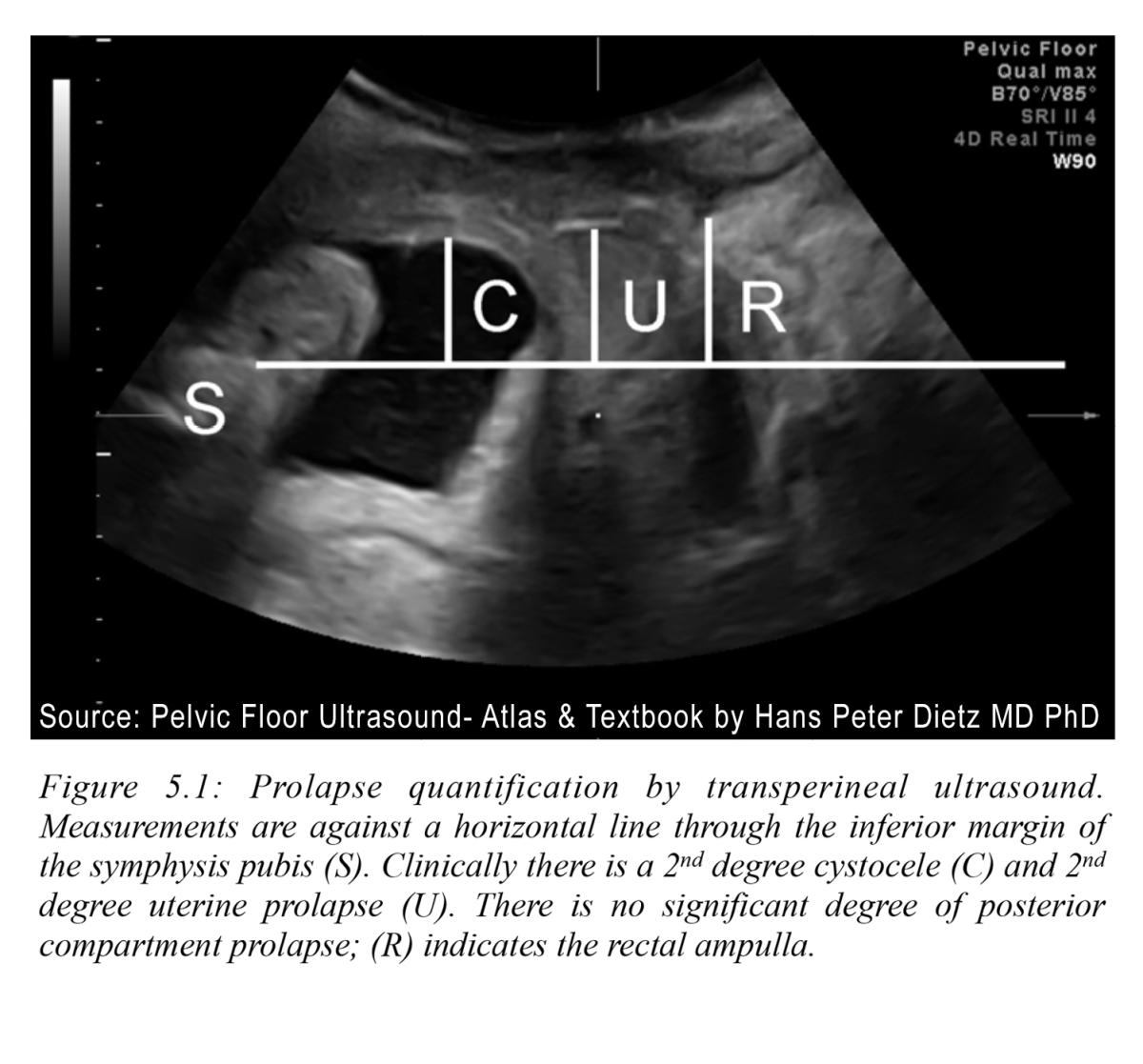
According to the most recent joint American College of Obstetricians and Gynecologists (ACOG)/American Urogynecologic Society (AUGS) statement, treatment is not recommended for asymptomatic pelvic organ prolapse, and ultrasound imaging is not included as part of the diagnostic evaluation — even for the 13% of U.S. women who eventually undergo surgery.6 Perhaps it is not surprising that, without in-depth evaluation of the underlying pelvic floor structures surgical repairs have a high failure rate — up to 30%.5 See Figure 5.1.
Their recommendation is to take a “wait and see” approach. But studies have shown that intervening sooner with pelvic floor muscle exercises can make a significant difference.7 With the number of women experiencing pelvic organ prolapse expected to increase by approximately 50% by 2050, it is time to rethink this outdated thinking. Routine evaluation with 3-D/4-D imaging can not only contribute to more precise diagnoses with pelvic floor prolapse,3 it can also be utilized to objectively measure results. With more precise and personalized intervention plans, we can potentially prevent progression over time.
Urinary Incontinence
Urinary incontinence affects up to 1 in 4 women, and this increases with age so that by age 65 up to 75% of women are affected.8 And yet, most women never seek help. In a study published in 2013, 95% of women reported a negative impact on their quality of life, but 64% of the women had never received any medical help.9 This is yet another missed opportunity to improve health and lives for women.
There are two main types of urinary incontinence: urgency and stress. Urgency incontinence, often related to detrusor muscle instability, occurs when there is involuntary urinary leakage related to an urge to void. Stress urinary incontinence is the leakage of urine associated with increased intraperitoneal pressure associated with physical activity, such as jumping, running or coughing.
Several factors have been implicated in the mechanisms underlying stress and urgency incontinence. These include damage to the pelvic floor, particularly those that lead to urethral hypermobility and decreased urethral sphincter tone, pelvic organ prolapse, alterations in functioning of the detrusor muscle, plus changes in the urothelium and urinary microbiome.10
Urgency incontinence is more common in older women, while the prevalence of stress incontinence peaks in the fifth decade of life. A significant percentage of women develop both, adding to the challenge in accurate diagnosis and treatment.
Ultrasound is helpful in ruling out other anatomical causes, including pelvic and bladder anomalies and masses. Using 3-D/4-D ultrasound can further enhance diagnostic accuracy by evaluating the pelvic floor structure, including the ligaments supporting the bladder and urethra, and urethral sphincter tone.3
Obstetrical Trauma
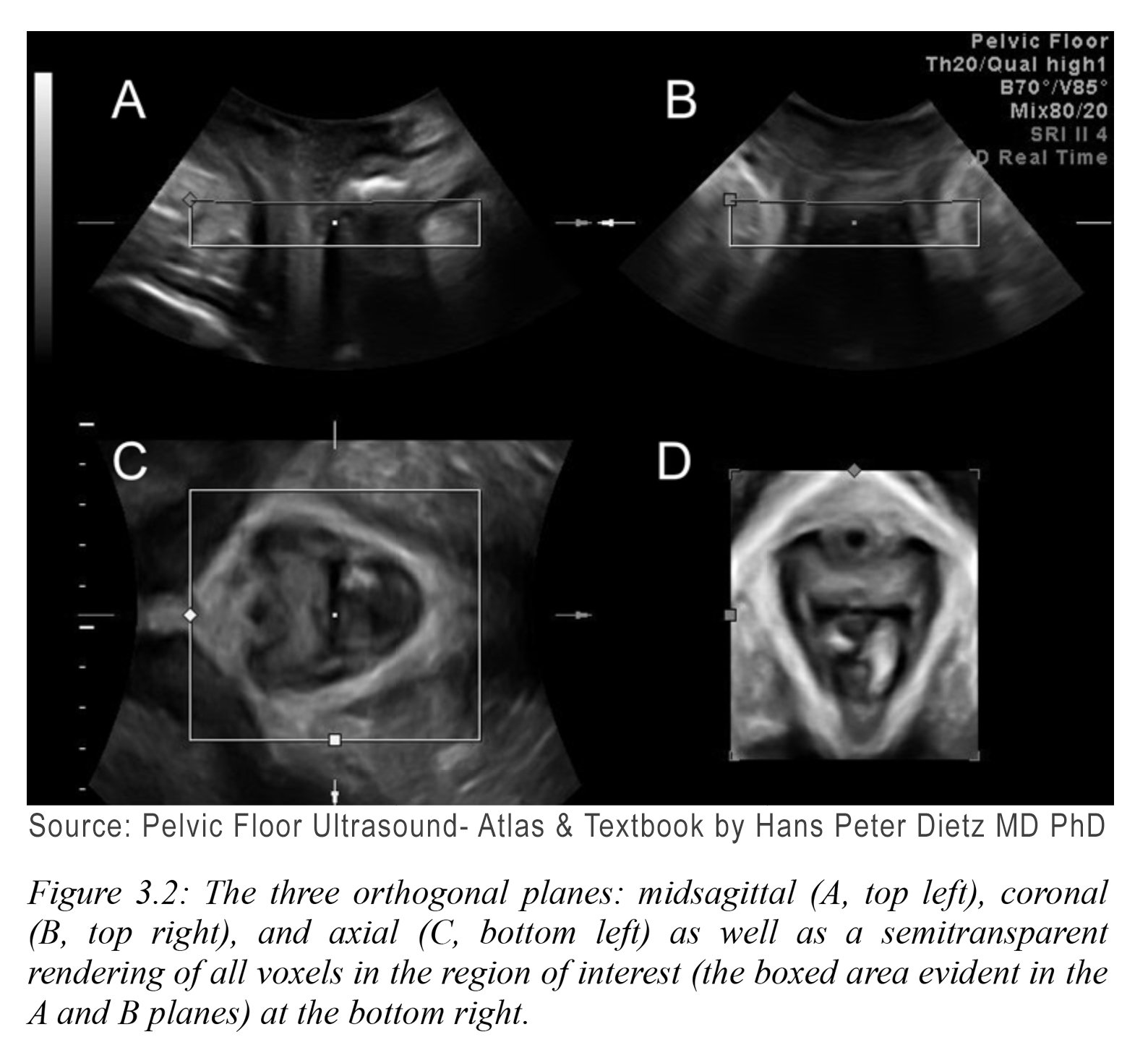
Trauma sustained during labor and vaginal delivery is one of the main contributors to pelvic floor disorders. While prolonged second stage labor, operative vaginal delivery and median episiotomy are well-established risk factors, recent studies have highlighted the role of apparently uncomplicated labor as well.11 Mechanisms contributing to pelvic floor disorders include mechanical injury to the pelvic floor soft tissues (muscle, connective tissue and nerves), ischemia and reperfusion injury and defective soft tissue remodeling.12 See Figure 3.2.
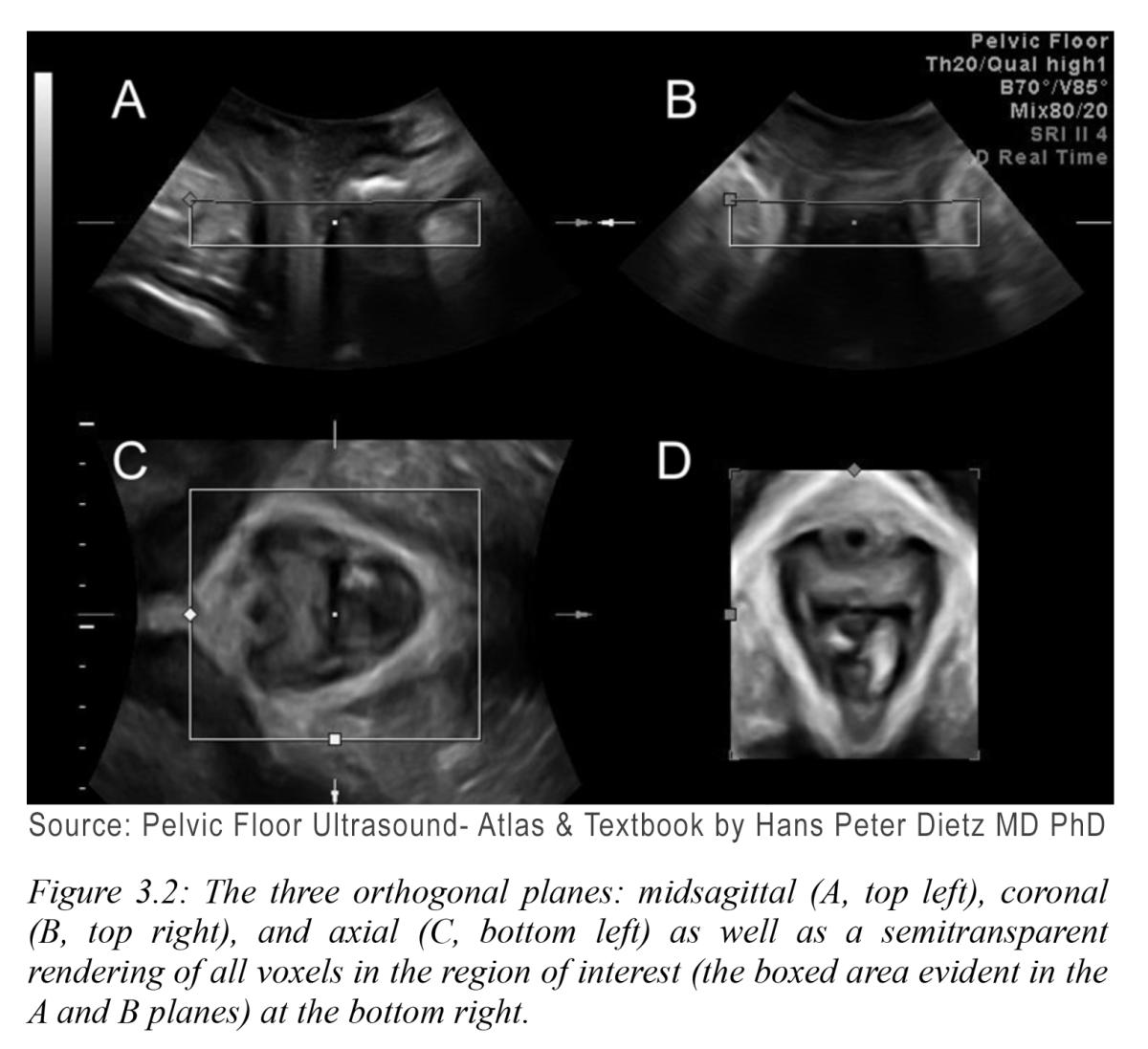
But perhaps the biggest contributor is that many injuries go unnoticed on physical exam and are left untreated. With the risk of pelvic floor disorders increasing with increasing age and parity, this opens a critical opportunity for improving diagnosis for earlier treatment.
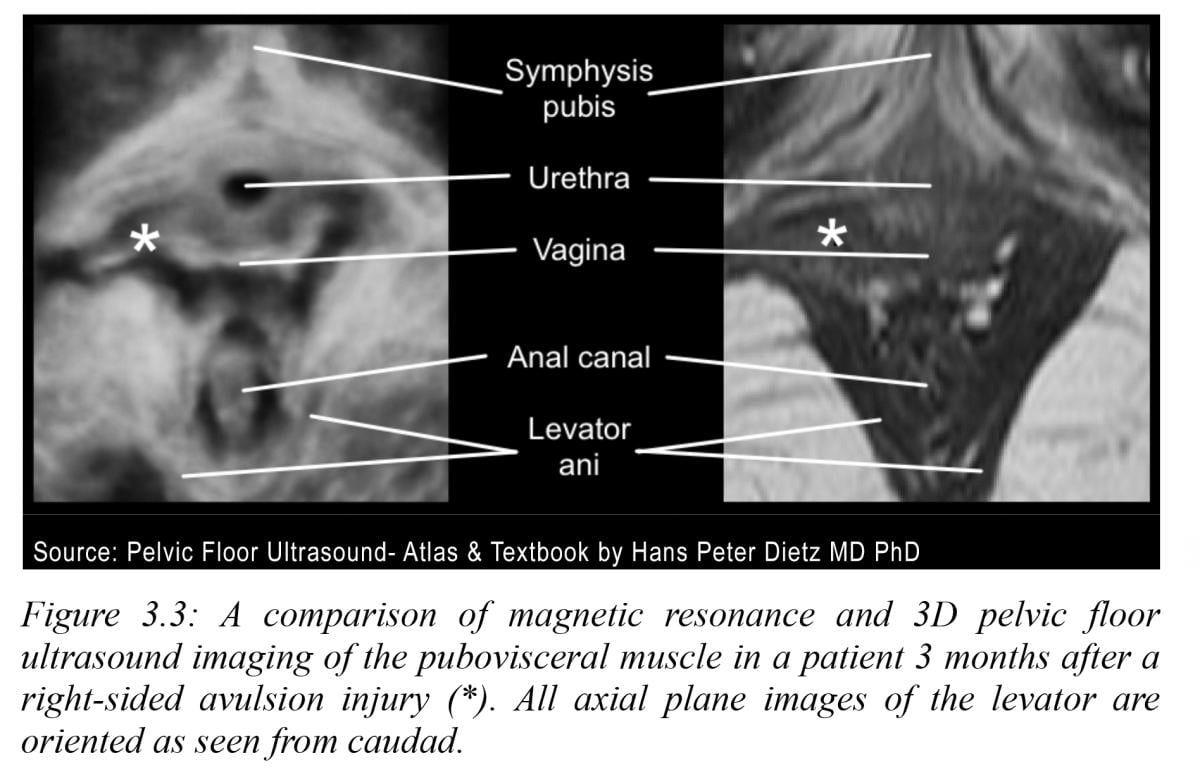
Ultrasound has been shown to be a far more effective modality than physical exam for detecting obstetrical trauma, particularly for anal sphincter injuries,13 levator ani and puborectalis muscle defects.14 It is generally acknowledged that standard X-rays and computed tomography (CT) scans offer limited value except in the case of retained foreign bodies. Studies show that soft tissue pelvic floor imaging with MRI is time consuming and offers inferior resolution of the critical anatomic structures as compared to high resolution 3-D/4-D dynamic ultrasound.15 Levator sling tears are visible as an echo poor dark gap in the echogenic white oval ring with this modality. See Figure 3.3.
SIDEBAR
Advantages of 3-D/4-D Imaging
By Robert L. Bard, MD, DABR, FASL
The increased application of pelvic reconstructive surgery with anterior – posterior repair and slings or mesh insertion has created its own set of new complications that add onto the postoperative trauma of routine vaginal deliveries and cesarean sections.
Translabial or transperineal ultrasound (TLU) is the best way to image the pelvic floor. It is not only cheaper and readily available when compared to MRI, but also able to provide real time images. Modern meshes are echogenic but invisible on X-ray or MRI. Four-dimensional imaging has been comprehensively prescribed for pelvic floor assessment, likelihood of pelvic floor prolapse, identifying placement of mesh and post-operative complications. In this article, we discuss the sonographic images that can be easily obtained in a clinical setting and used for informed treatment plans for patients.
Pain in the form of urethral and vaginal disorders has been reported but not well documented by current imaging modalities of MRI and TLU. Inflammatory hyperemia is optimally assessed by real time Doppler ultrasound technologies.17 Dietz et al have described modern 4-D ultrasound systems allowing visualization of the entire levator hiatus with levator ani, urethra, vagina, rectal ampulla, and anal canal imaged in the same plane and in real time, requiring very little transducer manipulation. This image is obtained by using 70-90 degree aperture and 80-90 degree acquisition angle. Pelvic meshes are echogenic polypropylene material easily identified with sonogram. To identify mesh length and thickness, we commonly used ultrasound systems equipped with a 5 MHz vaginal sector probe with an emission angle of at least 90 degrees. The mesh is identified in the midsagittal view and the distal-to-proximal length. Its thickness is measured and inflammation correlated with pain using qualitative and quantitative blood flow imaging technologies. Newer probes with 4-D capabilities include frequencies from 9-18 MHz providing surgical quality mapping information. Quantitative 4-D Doppler Histogram analysis demonstrates the pathologic inflammatory response and simultaneously quantifies the intensity of the disorder serving as a roadmap to intelligently following treatment progress. This allows an accurate assessment of the periurethral inflammatory tissue which aggravates pain and incontinence.18
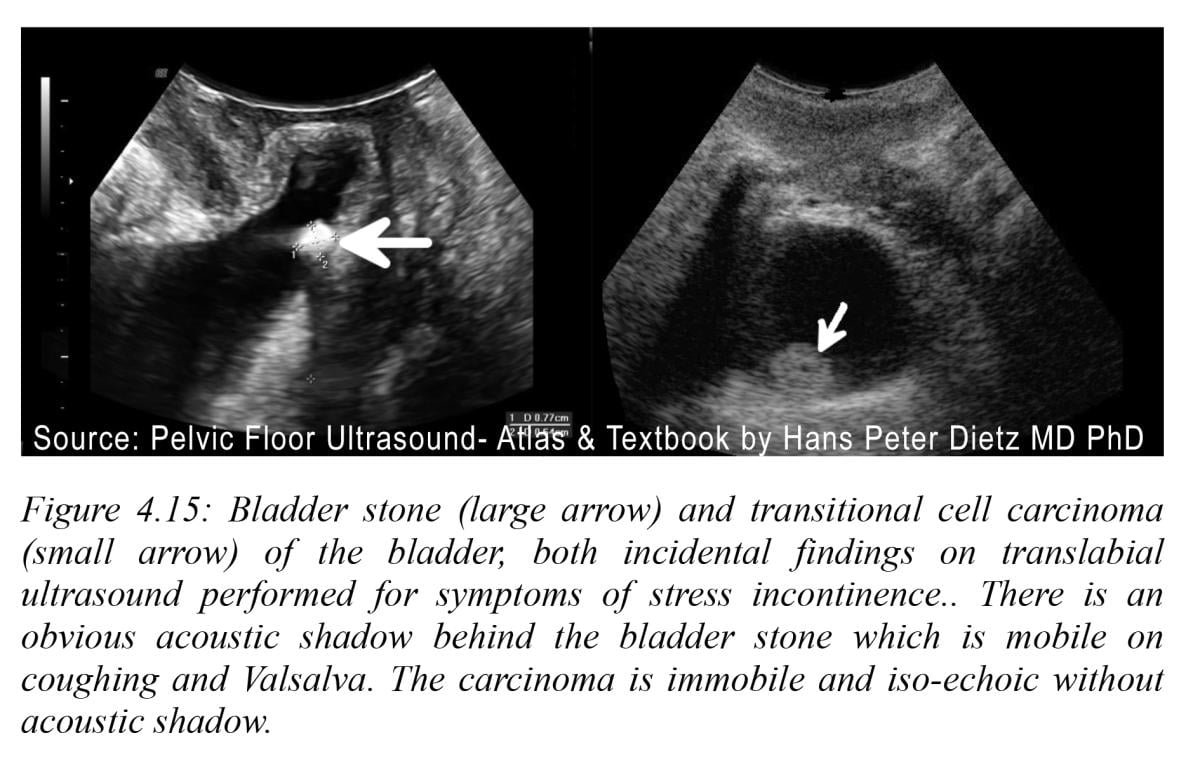
Inflammation that is chronic results in fibrosis and scar formation with tissue deformity and functional degradation due to nerve damage and involvement of adjacent tissues. Recent ultrasound breakthroughs in tissue elasticity with shear wave technologies quantify the area and degree of post inflammation effects. Real time dynamics of the urethra are optimally interpreted by the effect of Valsalva on the urethral mobility as a cause of SUI. Ultrasound permits onsite verification of urethral functionality as the distance between the bladder neck and the symphysis is readily documented. A recent study confirmed that the distances between the urethra and the symphysis pubis was significantly increased in the SUI group especially in the mid urethra. This suggests that focused treatment to the mid urethral segments may improve outcomes and attention diverted from the previous suspected bladder neck hypermobility. Highly echogenic bladder stones are white casting a sonic shadow and are distinguishable from less echogenic transitional cell carcinomas with Doppler neovascularity and accurately depicted tumor borders presented on the 3-D/4-D image reconstruction. See Figure 4.15.
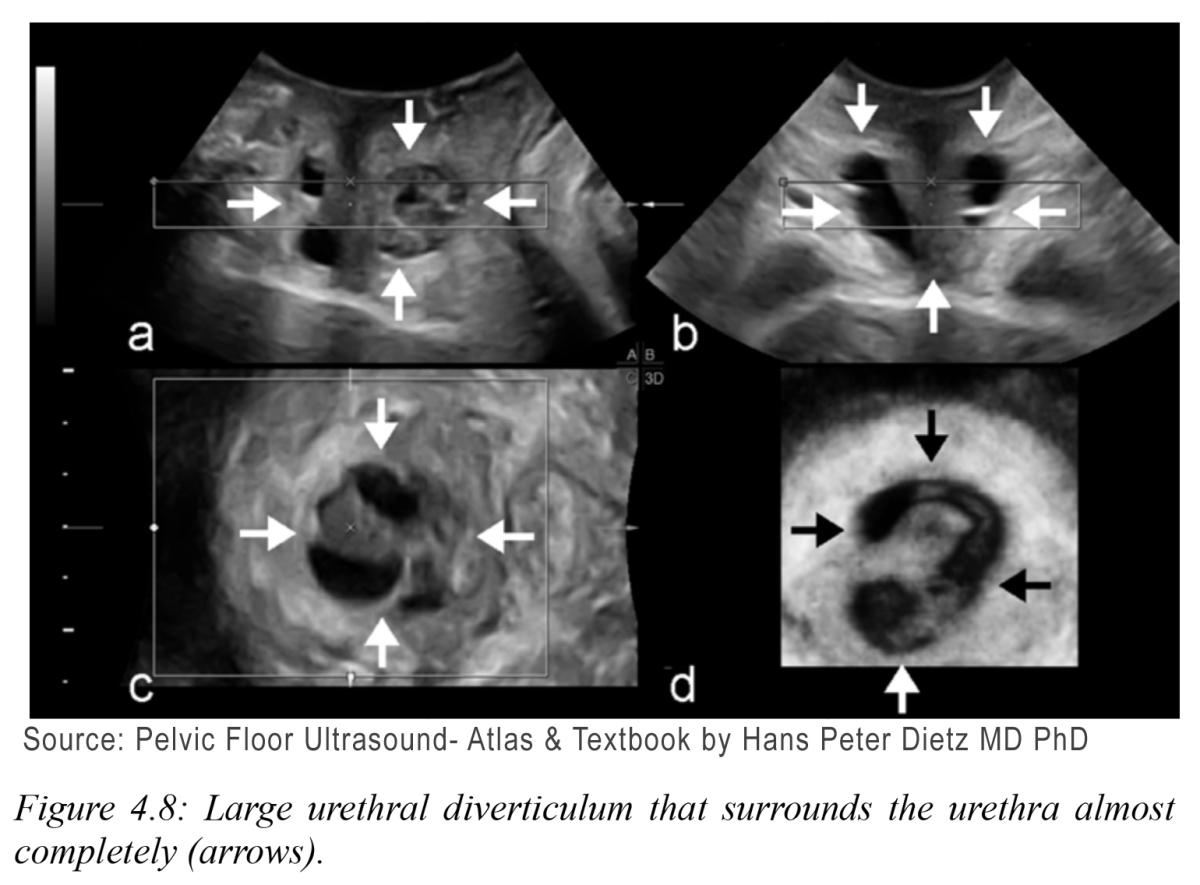
Cystic echo free urethral collections are easily visible in 2-D imaging as black areas. Three-dimensional mapping of complex cysts provides better surgical preoperative mapping. See Figure 4.8.
Use of ultrasound imaging in evaluation of mesh placement and complications is an essential component of patient workup when deciding treatment plans for patients. It is well described that excellent images of the pelvic floor can be obtained with ultrasound. In addition, mesh evaluation can be obtained to reduce risks of complications of surgery. It is a possible that ultrasound can be used to even identify patients that may not benefit from surgery. The future of ultrasound in mesh imaging may even identify how to improve surgical technique and mesh design.19-21
Furthermore, 3-D sonography and power Doppler angiography are techniques that may contribute new morphologic parameters in identifying mesh inflammation, urethritis and extrapelvic disease extension.22 It is clinically useful in the detection and grading coexistant bladder cancer and endometrial carcinoma. The addition of in vivo real time volume density measurements of inflammatory neovascularity may prove to be of value in determining the risk of mesh dislodgement noninvasively.
END SIDEBAR

Roberta Kline, M.D., is a board-certified ObGyn physician, Functional Genomics consultant, author and educator. At the forefront of advancing innovative solutions to improve women’s health, Kline has created and taught courses through faculty positions at the University of New England College of Osteopathic Medicine and The Graduate Institute. She was also instrumental in creating the Integrative and Functional Medicine program for a family practice residency program — the first of its kind when it launched in 2013. She has created clinical genomic testing and advanced personalized medicine training programs for clinicians and is co-author of Orthogenomics: Genome-Guided Therapies in Orthopedics.
Robert L. Bard, M.D., PC, DABR, FASLMS, is active medical director of Cancer Diagnostic Imaging Center (NYC), using advanced 3-D ultrasonographic Doppler imaging to detect cancers in breast, prostate, skin, thyroid, melanoma and other areas. He has extensive credentials as a clinical researcher/validator and has published medical textbooks and science journals. He is also a member of the ITN Editorial Advisory Board.
REFERENCES:
1. Nygaard I, Barber MD, Burgio KL, et al. Prevalence of symptomatic pelvic floor disorders in US women. JAMA. 2008; 300:1311–1316.
2. Faubion S et al. Recognition and Management of Nonrelaxing Pelvic Floor Dysfunction. Mayo Clin Proc. 2012 Feb; 87(2): 187–193.
3. Dietz HP Ultrasound imaging of the pelvic floor. Part II: three-dimensional or volume imaging. Ultrasound Obstet Gynecol 2004;23: 615 – 625
4. Chamié LP et al. Translabial US and Dynamic MR Imaging of the Pelvic Floor: Normal Anatomy and Dysfunction. 2018 RadioGraphics Vol. 38, No. 1.
5. ACOG Practice Bulletin Summary, Number 214. Pelvic Organ Prolapse. Obstetrics & Gynecology: November 2019 - Volume 134 - Issue 5 - p 1124-1127
6. ACOG/AUGS Guidance Update: Diagnosis and Management of Pelvic Organ Prolapse Including Role of Mesh Oct 28, 2019
7. Li C et al. The efficacy of pelvic floor muscle training for pelvic organ prolapse: a systematic review and meta-analysis. Int Urogynecol. 2016 Jul;27(7):981-92
8. Wu JM et al. Lifetime risk of stress urinary incontinence or pelvic organ prolapse surgery. Obstet Gynecol 2014;123:1201–6.
9. Sensoy N, Dogan N, Ozek B, Karaaslan L. Urinary incontinence in women: prevalence rates, risk factors and impact on quality of life. Pak J Med Sci 2013;29(3):818-822
10. Aoki Y et al. Urinary incontinence in women. Nat Rev Dis Primers. ; 3: 17042.
11. Rios, Gonzales et al. A Search for Novel Risk Factors for Obstetric Trauma. Obstetrics & Gynecology: May 2014 - Volume 123 - Issue - p 54S
12. Hafsa Memon and Victoria Handa Vaginal childbirth and pelvic floor disorders. Womens Health (Lond Engl). 2013 May ; 9(3.
13. Sultan A et al Anal-Sphincter Disruption during Vaginal Delivery N Engl J Med 1993;329:1905-11
14. Camila Carvalho de Araujo et al. Does vaginal delivery cause more damage to the pelvic floor than cesarean section as determined by 3D ultrasound evaluation? A systematic review. Review Int Urogynecol J. 2018 May;29(5):639-645.
15. Dietz HP Pelvic Floor Ultrasound: Atlas and Textbook 2016 HP DietzMD PhD FRANZCOG DDU CU
16. Zhao B, Wen L, Liu D etal 2022 J Ultrasound Med 41: 1439-1445
17. Schuettoff SA, Beyersdorff D, Gauruder-Burmester A, Tunn R.Visibility of the polypropylene tape after tension-free vaginal tape (TVT) procedure in women with stress urinary incontinence: comparison of introital ultrasound and magnetic resonance imaging in vitro and in vivo. Ultrasound Obstet Gynecol 2006; 27: 687–692.
18. Dietz HP, Wilson PD. The ‘iris effect’: how two-dimensional and three dimensional ultrasound can help us understand anti-incontinence procedures. Ultrasound Obstet Gynecol 2004; 23: 267–271.
19. Dietz HP, Barry C, Lim YN, Rane A. Two-dimensional and three-dimensional ultrasound imaging of suburethral slings. Ultrasound Obstet Gynecol 2005; 26: 175–179
20. Chantarasorn V, Shek KL, Dietz HP. Sonographic appearance of transobturator slings: implications for function and dysfunction. Int Urogynecol J 2011; 22: 493–498.
21. Fischer T, Ladurner R, Gangkofer A, Mussack T, Reiser M, Lienemann A. Functional cine MRI of the abdomen for the assessment of implanted synthetic mesh in patients after incisional hernia repair: initial results. Eur Radiol 2007; 17: 3123–3129.
22. Schuettoff S, Beyersdorff D, Gauruder-Burmester A, Tunn R. Visibility of the polypropylene tape after tension-free vaginal tape (TVT) procedure in women with stress urinary incontinence: comparison of introital ultrasound and magnetic resonance imaging in vitro and in vivo. Ultrasound Obstet Gynecol 2006; 27: 687–692.


 May 07, 2026
May 07, 2026 









