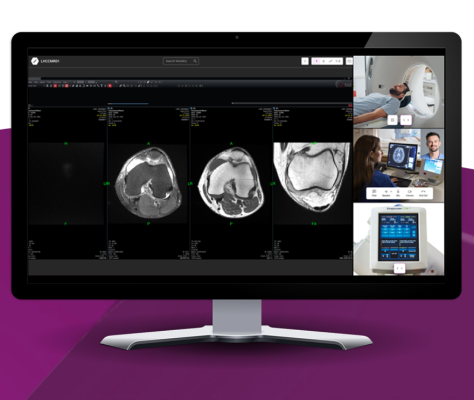
August 7, 2025 — DeepHealth, Inc., a wholly-owned subsidiary of RadNet, Inc., announced it has received FDA 510(k) clearance for TechLive – a remote scanning solution enabling centralized operation and supervision of MR, CT, PET/CT, and ultrasound procedures. Amid tech labor shortages and inflationary wage pressure, TechLive empowers technologists to scan for multiple locations, enables improved operational efficiency, extends center operating hours and enhances access to complex procedures. More than 300 of RadNet’s MR, CT, PET/CT, and ultrasound systems are now connected with DeepHealth’s TechLive, resulting in increased patient throughput and improved quality through remotely matching expert staff with complex modalities in real-time.
Over the past decade, radiology workloads have steadily increased, while the number of qualified technologists has not grown proportionally. In a pilot deployment at 64 locations inside of RadNet’s New York area facilities, TechLive significantly contributed to a 42% decrease in MRI room closure hours during the second quarter of 2025 compared with the same period in 2024, allowing more patients to receive timely scans.
Additionally, as a result of the remote expert involvement during the same pilot deployment, TechLive contributed to an increase in complex procedures. Furthermore, in extending remote scanning capabilities to Ultrasound, TechLive is enabling experienced senior sonographers and physicians to remotely guide technologists on-site through complex cases in a modality that is heavily dependent upon real-time, operator expertise.
“TechLive represents a paradigm shift in how we approach imaging operations,” said Sham Sokka, PhD, Chief Operating and Technology Officer at DeepHealth. “By enabling real-time remote expertise, we are not only addressing today’s staffing challenges, we are creating a foundation for more efficient, financially sustainable, and high-quality patient care across the broadest set of imaging modalities. This FDA clearance validates DeepHealth’s vision of a connected imaging ecosystem that scales human expertise beyond physical boundaries.”
Go to https://deephealth.com for more information.


 May 12, 2026
May 12, 2026 









