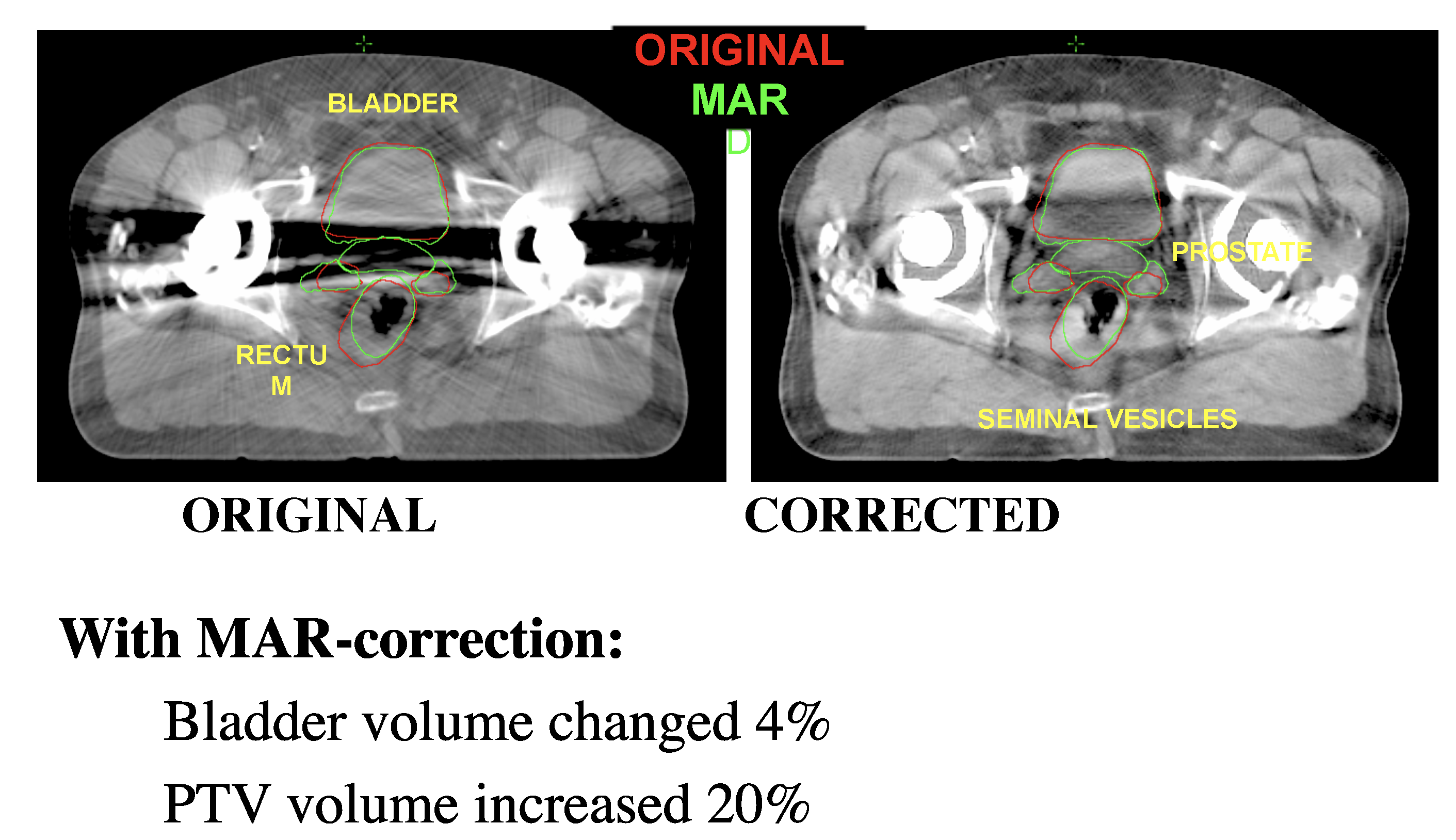
Metal artifact reduction led to a 20 percent increase in the planning target volume (PTV) in this prostate cancer case from Henry Ford Health System. The volume of the healthy bladder tissue to be spared was also reduced by 4 percent.
Computed tomography (CT) has long been the standard of care for imaging to plan radiation therapy (RT) treatments. CT offers several clinical advantages for treatment planning, including:
• Direct measurement of electron densities for radiation dose calculations;
• Clear visualization of high contrast bony landmarks;
• Excellent spatial fidelity; and
• Displays some soft tissue contrast for delineation.
However, CT imaging for radiotherapy planning offers its own set of challenges, not the least of which is it delivers extra radiation dose outside the target treatment area, and potential image artifacts. Carri Glide-Hurst, Ph.D., director of translational research for radiation oncology at Henry Ford Health System, led an ITN webinar sponsored by Philips Healthcare to address the newest advances in CT technology to enhance the modality’s usage for RT treatment planning.
Causes and Types of CT Image Artifacts
Imaging artifacts arise when there is a systematic difference between the reconstructed CT numbers and the true attenuation coefficients of the tissues being imaged. This results in part of the image being distorted or obscured.
CT scans can be impacted by a variety of image artifacts, including:
• Ring artifacts — caused by miscalibration of the scanner or failure of the detector element.
• Cupping — caused when polychromatic X-rays traverse through the body and become hardened, or a differential attenuation in the image.
• Streaking or banding — typically caused by bony anatomy and/or wide patient posture.
• Motion artifacts — caused by voluntary or involuntary movement of the patient during the scan.
• Metal artifacts — caused when the area being imaged has or is adjacent to a metallic object (implanted or external). The beam attenuation through
the metal is not well-approximated, according to Glide-Hurst, and the result is streaking, beam hardening or even a loss of information where the metal and
tissue interface.
Metal Artifact Reduction
Metal artifacts in particular can pose a challenge, especially for radiation oncology planning.
“This is something that’s increasing in importance as our subjects are becoming more aged,” said Glide-Hurst. “We’re treating patients into later years for their cancer radiation therapy, and also hip replacements and other kinds of implants are becoming more common for treatment of different ailments.”
The easiest way to solve the problem would be to remove the metal object, but this is rarely possible. Instead, most CT vendors have begun incorporating algorithms for metal artifact reduction into their systems or as add-ons.
Glide-Hurst reviewed several of the current CT technologies for metal artifact reduction:
• Metal Artifact Reduction (MAR) algorithm, Siemens — This algorithm thresholds metal objects with 3,000 Hounsfield units (HU) and identifies projections from unreliable regions of the image. MAR then modifies the raw projection data that is used with filtered back projection (FBP).
• Single energy metal artifact reduction (SEMAR), Canon/Toshiba — On Canon CT systems, the metal is identified in the raw CT data (also known as a sinogram), and then trace projections are used to correct the raw data and reconstruct it.
• Metal artifact reduction for orthopedic implants (O-MAR), Philips — Philips’ algorithm thresholds the image to generate a metal-only image, then
interpolates the result in a tissue classification scheme. Several sinograms are forward projected into an error sinogram to create a masked sinogram (showing the error masked by the metal). The masked sinogram is put through one last FBP to create a corrected image.
• Adaptive filtration method — While not specifically designed for metal artifact reduction, adaptive filtration is a common feature on most CT scanners, according to Glide-Hurst. This technique smooths the attenuation profile and then reconstructs the image to reduce streaking in photon-starved images.
Optimizing Imaging Dose in RT Planning
Glide-Hurst said she has fielded questions on whether CT imaging dose matters in radiotherapy planning when the patient is already receiving such large therapy doses. “The thing you have to think about is imaging dose is not localized compared to our treatment beams, which are really tailored to patient’s tumor and anatomy,” she explained. Using CT and/or 4-D CT exposes organs outside the target treatment area to concomitant radiation dose. Glide-Hurst added that 4-D CT is more widely used today for advanced applications like adaptive radiation therapy and re-simulation, and in radiosensitive organs such as the breast.
Recent efforts in radiology circles to reduce imaging dose have included concepts like Image Wisely and the As Low As Reasonably Achievable (ALARA) principle. Applying these concepts to imaging for radiotherapy treatment planning, however, is not as straightforward. “The one thing you have to keep in mind is the needs are a little bit different,” said Glide-Hurst. She explained that wider bores and fields of view are often needed for RT imaging because of the immobilization devices and different patient stances required.
Ultimately, while the implementation is slightly different, the thinking is virtually the same, according to Glide-Hurst: The risks and benefits needed to be assessed, but it is critical to remember that imaging can greatly improve the therapeutic ratio for radiotherapy treatment.
CT Dose Reduction Technologies in Treatment Planning
In the webinar, Glide-Hurst discussed several current and emerging technologies to help optimize imaging dose for radiotherapy treatment planning.
Filtered back projection (FBP) has been a gold-standard method for improving image quality in radiology. However, Glide-Hurst noted that as low-dose protocols are enacted, FBP can demonstrate increased noise and image artifacts, and decrease the contrast-to-noise ratio (CNR).
“So the question is how can we lower doses without compromising image quality?” she said.
One method that many CT vendors now offer is iterative reconstruction algorithms. The technology has become commonplace in radiology, but its potential in radiation oncology is still being explored.
Henry Ford Health System tested an iterative reconstruction model that generates synthesized projections by modeling CT data collection via statistics and the optical component of the detectors themselves. During the initial validation, the research team focused on two areas:
• Concerns related to image quality included spatial resolution, low-contrast resolution and uniformity; and
• Treatment planning concerns focused on the CT-electron density, delineation and dosimetry.
The patient population included female high dose rate brachytherapy (HDR) patients who were imaged daily for treatment verification. “We thought this would be an optimal population to study because they were already undergoing 3-5 imaging sessions per person, and we could gradually reduce the dose while we do multiple reconstructions,” said Glide-Hurst. Image data was graded against the reference dose on a scale of 1 to 6 — 1 meant the iteratively reconstructed image was better than reference, 3 indicated they were equal, and 6 indicated unacceptable image quality. Results showed that iterative reconstruction was significantly better than filtered back projection in terms of noise levels and low-contrast interface.
Combining Technologies
Henry Ford conducted another series of studies to explore how metal artifact reduction and dose optimization together impacted treatment planning. The primary objective was trying to improve contouring of the treatment dose in 11 bladder or prostate cancer patients with bilateral hip replacements. A single physician delineated the original (VO) and artifact corrected (VAC) CT data sets, and then a dice similarity coefficient (DSC) and overlap index (OI) were used to determine the difference in contouring between the two sets.
In one bladder carcinoma case, the researchers found the bladder volume was overestimated on the original scan by more than 30 percent. In one prostate cancer case, MAR correction led to a 4 percent reduction in the healthy bladder tissue volume and a 20 percent increase in the planning target volume (PTV). “Had we not had the ability to visualize that anatomy in that prostate, we could have compromised this patient’s outcome,” said Glide-Hurst.
In the final results, 9 of the 11 patients had their minimum target dose significantly reduced. “That’s an important result because that shows that it really influenced what the target volumes were going to be in these pelvic cases,” Glide-Hurst explained. Furthermore, the metal artifact reduction successfully revealed bladder and rectal status, and the organ-at-risk (OAR) volume change ranged from -32.4 to 31 percent.
Next-Generation Technologies
Glide-Hurst concluded the webinar by looking into the future at emerging CT applications in radiation oncology.
While iterative reconstruction has proven beneficial in improving image quality at low doses, some are already looking at its successor, model-based reconstruction (MBR), as an even better solution. Glide-Hurst said MBR is similar to iterative reconstruction except that it adds a combination of artificial object data and projections of the raw data into the reconstruction. “This gives you the ability to highlight certain tissues because you can change a bit of the weighting,” she said. MBR provides significant noise reduction compared to filtered back projection.
She noted that adding more detector rows to the CT scanner also improves image quality for treatment planning. More rows increases the detector coverage area and reduces the scan time while still producing high-resolution images. Drawbacks of adding detector rows include increased cost for CT equipment as well as potential issues with table movements seen in some reconstruction models.
According to Glide-Hurst, dual-energy CT acquisition is also gaining operational traction in radiation oncology. Multiple CT vendors now offer dual-energy scanning, where objects are scanned at both 80 and 140 kVp to better assess tissue composition.
Glide-Hurst said the University of Wisconsin-Madison has been using dual-energy CT (DECT) for multiple applications throughout its radiation oncology department. For one example, using virtual non-contrast images generated through DECT may eliminate the need for an additional non-contrast scan during treatment planning, minimizing the imaging dose to the patient. In addition, the information collected from the DECT scan can improve the accuracy of therapeutic dose calculations, particularly for proton therapy and low-energy brachytherapy.
“They also think it may offer opportunities for improved visualization and quantification of iodine uptake for tumor staging, delineation and characterization,” concluded Glide-Hurst.


 April 30, 2026
April 30, 2026 









