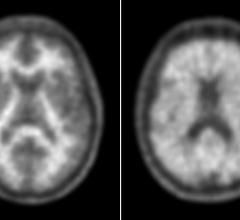
Satoshi Minoshima, M.D., Ph.D., chairman of the Society of Nuclear Medicine and Molecular Imaging Scientific Committee ...
Elekta and Royal Philips announced that The Institute of Cancer Research, London, a world-leading cancer research institution, working with its clinical partner The Royal Marsden NHS Foundation Trust, will join the Elekta MR Linac Research Consortium, a group with a mission to develop an integrated magnetic resonance imaging (MRI)-guided radiation therapy system.
A simple noninvasive blood test matched with state-of-the-art molecular imaging of individual cells could help oncologists understand their patients’ chances of survival, said researchers at the Society of Nuclear Medicine and Molecular Imaging’s 2014 Annual Meeting (SNMMI).
AT A GLANCE Organization: Expert Radiology Management Services, LLC Specialty: Subspecialty teleradiology — neuro and ...
June 18, 2014 — Researchers at Sandia National Laboratories, along with collaborators from Rice University and the Tokyo Institute of Technology, are developing new terahertz detectors based on carbon nanotubes that could lead to significant improvements in medical imaging, airport passenger screening, food inspection and other applications.
June 18, 2014 — A new molecular imaging agent has been developed to help clinicians find as much cancer as possible, whether it is responding favorably or not, in an effort to improve clinical decision making for prostate cancer patients, said researchers at the 2014 annual meeting of the Society of Nuclear Medicine and Molecular Imaging (SNMMI).
June 17, 2014 — Navidea Biopharmaceuticals Inc. announced results from a post-hoc analysis of patient data from the company’s phase III clinical trial (NEO3-06) of Lymphoseek in head and neck cancer.
Radiology departments have many different needs and face a wide variety of challenges that can impact their departments ...
June 17, 2014 — A total of 16 pioneering medical innovations were showcased in front of approximately 4,000 healthcare providers and experts at Premier Inc.'s sixth annual Innovation Celebration, such as the world's first and only intravenous implantable cardiac defibrillator (ICD).
June 16, 2014 — Piramal Imaging released details from research presented at the 2014 annual meeting of the Society of Nuclear Medicine and Molecular Imaging (SNMMI) in St. Louis. Research findings relating to the development and approval of Neuraceq (florbetaben F18 injection) were presented during the AD II: Advanced Approaches Session.
In the last 30 years, since mammography was introduced, late-stage breast cancer incidence has decreased by 37 percent, a new study from the University of Michigan Comprehensive Cancer Center finds.
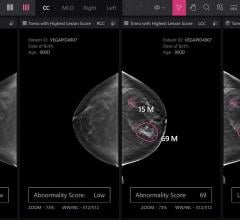
Despite decades of progress in breast imaging, one challenge continues to test even the most skilled radiologists ...
June 16, 2014 — Carestream’s Smart Link helps ensure optimal system performance and uptime by continuously monitoring operations of its healthcare IT and digital imaging systems at healthcare providers across the globe. Smart Link is provided as part of Carestream’s customer service maintenance agreements.
June 16, 2014 — Children with heart disease are exposed to low levels of radiation during X-rays, which do not significantly raise their lifetime cancer risk. However, children who undergo repeated complex imaging tests that deliver higher doses of radiation may have a slightly increased lifetime risk of cancer, according to researchers at Duke Medicine. The findings, published June 9 in the American Heart Association (AHA) journal Circulation, represent the largest study of cumulative radiation doses in children with heart disease and associated predictions of lifetime cancer risk.
Varian Medical Systems introduced the ProBeam Compact proton therapy system at the annual meeting of the Particle Therapy Co-Operative Group (PTCOG 53).
Bayer Radiology’s Barbara Ruhland and Thom Kinst discuss how radiology departments can address the many different ...
Elsevier, a world-leading provider of scientific, technical and medical information products and services, announced today the acquisition of Amirsys.
With financial constraints continuing to affect healthcare providers, there is a growing need for organizations to explore new means to generate revenue. For many imaging enterprises, the answer may lie in expanding their breast imaging services and incorporating leading-edge technologies like digital breast tomosynthesis – which can be done easier than previously believed. This is due to recently introduced software solutions, like Intelerad’s Tomosynthesis Module, which integrates directly into InteleViewer, thus providing the diagnostic tools that radiologists require and eliminating the need for costly workstations dedicated to the modality.
See how standardizing on Siemens' breast imaging solutions has helped Heritage Valley Health System optimize efficiency ...
June 13, 2014 — The U.S. Food and Drug Administration (FDA) today approved a new use for Navidea Biopharmaceuticals’ Lymphoseek (technetium Tc-99m tilmanocept) injection, a radioactive diagnostic imaging agent used to help doctors determine the extent a type of cancer called squamous cell carcinoma has spread in the body’s head and neck region.
aycan is now listed as a manufacturer on NASA’s Solutions for Enterprise-Wide Procurement (SEWP) list through business partner and contract holder Alvarez & Associates.
For years researchers have been developing molecular imaging techniques that visualize hormonally active breast cancer cells — specifically those testing positive for human epidermal growth factor receptor 2 (HER2). A recent innovation in breast cancer biomarkers seeks the HER3 receptor instead, which could mean more comprehensive breast cancer imaging and potential treatments, say experts presenting data during the Society of Nuclear Medicine and Molecular Imaging’s 2014 Annual Meeting (SNMMI).
June 12, 2014 — A recent peer-reviewed article published earlier this year in the British Medical Journal (BMJ) by Steve Brozak and Anne Marie Noronha again establishes a case for an alternative to the conventional endoscope cleaning process and reviews why conventional cleaning puts patients at risk of infection.
June 12, 2014 — Philips Healthcare recently introduced Vereos PET/CT, the first digital PET/CT (positron emission tomography/computed tomography) scanner, at the 2014 annual meeting of the Society of Nuclear Medicine and Molecular Imaging (SNMMI) in St. Louis. In addition to Vereos, Philips showcased a selection of molecular imaging solutions designed to deliver high image quality, critical clinical information and greater connectivity.


 June 19, 2014
June 19, 2014